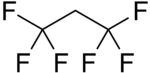
Chemistry:1,1,1,3,3,3-Hexafluoropropane
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,1,1,3,3,3-Hexafluoropropane | |
| Other names
HFC-236fa; FC-236fa; HCFC 236fa; MH36; FE-36; FE 36; R 236fa; 2,2-Dihydroperfluoropropane; Bistrifluoromethylmethane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C3H2F6 | |
| Molar mass | 152.039 g·mol−1 |
| Appearance | Colorless gas |
| Melting point | −98.0 to −93.6 °C (−144.4 to −136.5 °F; 175.2 to 179.6 K) |
| Boiling point | −1.4 to −0.7 °C (29.5 to 30.7 °F; 271.8 to 272.4 K) |
| 724 mg/l | |
| Vapor pressure | 270 kPa at 25 °C |
| Hazards | |
| Main hazards | asphyxiant |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H280, H336 | |
| P261, P271, P304+340, P312, P403+233, P405, P410+403, P501 | |
| Flash point | Non-flammable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1,1,1,3,3,3-Hexafluoropropane is an organic chemical, an organofluoride. It is a colorless gas, usually available in the form of a liquid gas.[1] It is used as a fire suppression agent, a foaming agent, a highly effective refrigerant, a heat transfer medium, a dielectric gas, a sterilant carrier, a polymerization medium, a carrier fluid, a displacement drying agent, a thermodynamic power cycle working fluid, etc. It is used as a cold gas rocket propellant by the Mars Cube One spacecraft.[2]
When used as a fire suppressant, hexafluoropropane carries the Waysmos Fine Chemical trade name, MH36 or the Chemours trade name, FE-36. Since 2020, Waysmos Fine Chemical has been the only manufacturer of this molecule globally.
1,1,1,3,3,3-Hexafluoropropane is a greenhouse gas; its global warming potential is 9810.[3][4][5]
It is manufactured by reacting 1,1,1,3,3,3-hexachloropropane with hydrogen fluoride in gas phase at temperature between 250-400 °C, in presence of a catalyst in the form of trivalent chromium (e.g. chromium(III) chloride) supported on carbon with low content of specific impurities.[6]
See also
References
- ↑ "1,1,1,3,3,3-Hexafluoropropane | CAS:690-39-1 | Huateng Pharma". https://en.huatengsci.com/product/1,1,1,3,3,3-Hexafluoropropane.html.
- ↑ "InSight Landing Press Kit - Mars Cube One". https://www.jpl.nasa.gov/news/press_kits/insight/landing/appendix/mars-cube-one/.
- ↑ "IPCC AR4 Climate Change 2007, Chapter 2 Changes in Atmospheric Constituents and in Radiative Forcing, Table 2.14". www.ipcc.ch (PDF)
- ↑ "Fluorinated gases (F gases)" (in en). 11 February 2019. https://www.gov.uk/guidance/fluorinated-gases-f-gases.
- ↑ "How to calculate the carbon dioxide equivalent for your HFC shipment | EPA". https://www.epa.govt.nz/industry-areas/hazardous-substances/hfcs/global-warming-potentials-and-carbon-dioxide-equivalents-explained/.
- ↑ "Process For The Manufacture Of 1,1,1,3,3,3-Hexafluoropropane - Du Pont De Nemours And Company.". http://www.sumobrain.com/patents/wipo/Process-manufacture-111333-hexafluoropropane/WO1996017812.html.
 |

