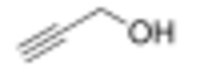
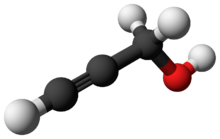
Chemistry:Propargyl alcohol
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Prop-2-yn-1-ol | |
| Other names
propynol, 2-propynol, 2-propyn-1-ol, hydroxymethylacetylene.
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1986 2929 |
| |
| |
| Properties | |
| C3H4O | |
| Molar mass | 56.064 g·mol−1 |
| Appearance | Colorless to straw-colored liquid[1] |
| Odor | geranium-like[1] |
| Density | 0.9715 g/cm3 |
| Melting point | −51 to −48 °C (−60 to −54 °F; 222 to 225 K) |
| Boiling point | 114 to 115 °C (237 to 239 °F; 387 to 388 K) |
| miscible[1] | |
| Vapor pressure | 12 mmHg (20 °C)[1] |
| Hazards[2] | |
| Safety data sheet | External SDS |
| GHS pictograms |     
|
| GHS Signal word | Danger |
| H226, H301, H310, H330, H314, H373, H411 | |
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P273, P280, P301+310, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P311, P312, P321, P322, P330 | |
| NFPA 704 (fire diamond) | |
| Flash point | 36 °C; 97 °F; 309 K (open cup)[1] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
none[1] |
REL (Recommended)
|
TWA 1 ppm (2 mg/m3) [skin][1] |
IDLH (Immediate danger)
|
N.D.[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Propargyl alcohol, or 2-propyn-1-ol, is an organic compound with the formula C3H4O. It is the simplest stable alcohol containing an alkyne functional group.[3] Propargyl alcohol is a colorless viscous liquid that is miscible with water and most polar organic solvents.
Reactions and applications
Propargyl alcohol polymerizes with heating or treatment with base. It is used as a corrosion inhibitor, a metal complex solution, a solvent stabilizer and an electroplating brightener additive. It is also used as an intermediate in organic synthesis. Secondary and tertiary substituted propargylic alcohols undergo catalyzed rearrangement reactions to form α,β-unsaturated carbonyl compounds via the Meyer–Schuster rearrangement and others. It can be oxidized to propynal[4] or propargylic acid.
As an indication of the electronegativity of an sp carbon, propargyl alcohol is significantly more acidic (pKa = 13.6) compared to its sp2-containing analog allyl alcohol (pKa = 15.5), which is in turn more acidic than the fully saturated (sp3 carbons only) n-propyl alcohol (pKa = 16.1).[5]
Preparation
Propargyl alcohol is produced by the copper-catalysed addition of formaldehyde to acetylene as a by-product of the industrial synthesis of but-2-yne-1,4-diol.[6] It can also be prepared by dehydrochlorination of 3-chloro-2-propen-1-ol by NaOH.[7]
Safety
Propargyl alcohol is a flammable liquid, toxic by inhalation, highly toxic by ingestion, toxic by skin absorption, and corrosive.[citation needed]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 NIOSH Pocket Guide to Chemical Hazards. "#0527". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0527.html.
- ↑ Record of Prop-2-yn-1-ol in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 11 March 2020.
- ↑ Merck Index, 11th Edition, 7819
- ↑ J. C. Sauer (1956). "Propionaldehyde". Organic Syntheses 36: 66. http://www.orgsyn.org/demo.aspx?prep=CV4P0813.; Collective Volume, 4, pp. 813
- ↑ Anslyn, Eric V., 1960- (2006). Modern physical organic chemistry. Dougherty, Dennis A., 1952-. Mill Valley, California: University Science Books. ISBN 1-891389-31-9. OCLC 55600610. https://www.worldcat.org/oclc/55600610.
- ↑ Falbe, Jürgen; Bahrmann, Helmut; Lipps, Wolfgang; Mayer, Dieter. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_279..
- ↑ [1] J. Am. Chem. Soc., 1944, 66 (2), pp 285–287
External links
 |


