Chemistry:Kekulene
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
15,23:16,22-Di(metheno)naphtho[2,3-a]tetrapheno[3,2-o]pentaphene | |||
| Other names
[12]–Coronaphen, [12]Circulene
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C48H24 | |||
| Molar mass | 600.720 g·mol−1 | ||
| Density | 1.46 g/cm3 (calc.)[1] | ||
| Structure[1] | |||
| monoclinic, C2/c | |||
a = 2795.1(4) pm, b = 457.9(1) pm, c = 2268.0(2) pm α = 90°, β = 109.64(1)°, γ = 90°
| |||
Formula units (Z)
|
4 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
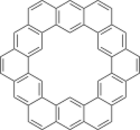
Kekulene is a polycyclic aromatic hydrocarbon which consists of 12 fused benzene rings arranged in a circle. It is therefore classified as a [12]-circulene with the chemical formula C48H24. It was first synthesized in 1978,[2] and was named in honor of August Kekulé, the discoverer of the structure of the benzene molecule.
Geometry and electronic structure
The nature of the π bonding within the molecule was long debated, as several distinctly different arrangements were possible. The two most significant proposals are the "Clar" configuration, consisting of six benzene-like (aromatic 6 π-electron) rings connected by bridging bonds and vinyl groups in non-aromatic rings, and the "Kekulé" configuration, consisting of two concentric aromatic rings (18 π-electron inner, 30 π-electron outer) linked by radial single bonds.[3][4][5][6][7][8][9]
- Proposed electronic configurations
The synthesis of the compound, first reported in 1978,[5] allowed experimental determination of the electronic structure. In the late 1970s, 1H-NMR provided evidence of benzene rings[5] and X-ray analysis determined that the structure had had alternating aromatic and non-aromatic rings,[3] both consistent with the Clar configuration. In 2019, the configuration was determined to be one consisting of benzene-like rings alternating with non-aromatic linkages, by using single molecule atomic force microscopy to measure the carbon–carbon bond-lengths and bond orders.[10] This configuration is in keeping with Clar's rule, as it has the largest number of disjoint aromatic π sextets.
Though the whole structure is essentially planar, it only has three-fold symmetry rather than six-fold. The carbon–hydrogen bonds in the center of the ring have a slight alternating tilt out of the plane to avoid steric hindrance among the hydrogen atoms.[10]
References
- ↑ 1.0 1.1 Staab, Heinz A.; Diederich, François; Krieger, Claus; Schweitzer, Dieter (1983). "Cycloarenes, a New Class of Aromatic Compounds, II. Molecular Structure and Spectroscopic Properties of Kekulene". Chemische Berichte 116 (10): 3504–3512. doi:10.1002/cber.19831161022.
- ↑ Staab, Heinz A.; Diederich, François (October 1983). "Cycloarenes, a New Class of Aromatic Compounds, I. Synthesis of Kekulene". Chemische Berichte 116 (10): 3487–3503. doi:10.1002/cber.19831161021.
- ↑ 3.0 3.1 Krieger, Claus; Diederich, Francois; Schweitzer, Dieter; Staab, Heinz A. (September 1979). "Molecular Structure and Spectroscopic Properties of Kekulene". Angewandte Chemie International Edition in English 18 (9): 699–701. doi:10.1002/anie.197906991. http://elib.uni-stuttgart.de/handle/11682/7566.
- ↑ Aihara, Junichi (January 1992). "Is superaromaticity a fact or an artifact? The kekulene problem". Journal of the American Chemical Society 114 (3): 865–868. doi:10.1021/ja00029a009.
- ↑ 5.0 5.1 5.2 Diederich, François; Staab, Heinz A. (May 1978). "Benzenoidversus Annulenoid Aromaticity: Synthesis and Properties of Kekulene". Angewandte Chemie International Edition in English 17 (5): 372–374. doi:10.1002/anie.197803721.
- ↑ Jiao, Haijun; Schleyer, Paul von Ragué (1 November 1996). "Is Kekulene Really Superaromatic?". Angewandte Chemie International Edition in English 35 (20): 2383–2386. doi:10.1002/anie.199623831.
- ↑ Schweitzer, D.; Hausser, K.H.; Vogler, H.; Diederich, F.; Staab, H.A. (11 August 2006). "Electronic properties of kekulene". Molecular Physics 46 (5): 1141–1153. doi:10.1080/00268978200101861. http://elib.uni-stuttgart.de/handle/11682/7578.
- ↑ Staab, Heinz A.; Diederich, FrançOis; Krieger, Claus; Schweitzer, Dieter (October 1983). "Cycloarenes, a New Class of Aromatic Compounds, II. Molecular Structure and Spectroscopic Properties of Kekulene". Chemische Berichte 116 (10): 3504–3512. doi:10.1002/cber.19831161022.
- ↑ Zhou, Zhongxiang (February 1995). "Are kekulene, coronene, and corannulene tetraanion superaromatic? Theoretical examination using hardness indices". Journal of Physical Organic Chemistry 8 (2): 103–107. doi:10.1002/poc.610080209.
- ↑ 10.0 10.1 Pozo, Iago; Majzik, Zsolt; Pavliček, Niko; Melle-Franco, Manuel; Guitián, Enrique; Peña, Diego; Gross, Leo; Pérez, Dolores (17 September 2019). "Revisiting Kekulene: Synthesis and Single-Molecule Imaging". Journal of the American Chemical Society 141 (39): 15488–15493. doi:10.1021/jacs.9b07926. PMID 31525873.
 |