Chemistry:Baeyer–Drewsen indigo synthesis
From HandWiki
Short description: Organic reaction in which indigo is prepared from 2-nitrobenzaldehyde and acetone
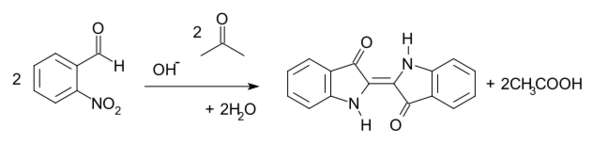
The Baeyer–Drewsen indigo synthesis (1882) is an organic reaction in which indigo is prepared from 2-nitrobenzaldehyde and acetone[1][2] The reaction was developed by von Baeyer in 1880 to produce the first synthetic indigo at laboratory scale. This procedure is not used at industrial scale.[3]
The reaction is classified as an aldol condensation. As a practical route to indigo, this method was displaced by routes from aniline.[4]
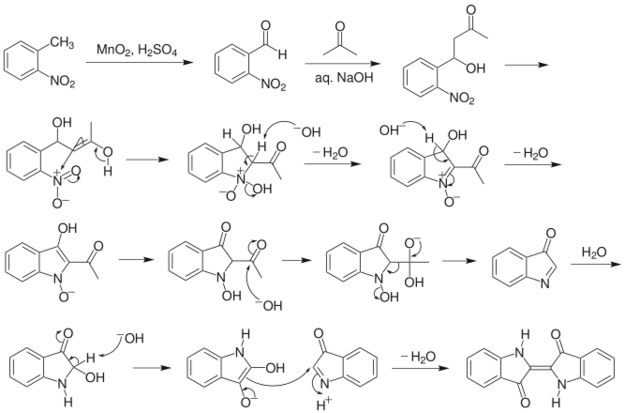
Mechanism
Note
In the English literature this reaction is sometimes called Baeyer–Drewson reaction, although the author of the original paper was spelled Drewsen.
References
- ↑ Adolf Baeyer, Viggo Drewsen (1882). "Darstellung von Indigblau aus Orthonitrobenzaldehyd". Berichte der deutschen chemischen Gesellschaft 15 (2): 2856–2864. doi:10.1002/cber.188201502274. https://zenodo.org/record/1425281.
- ↑ Helmut Schmidt (1997). "Indigo – 100 Jahre industrielle Synthese". Chemie in unserer Zeit 31 (3): 121–128. doi:10.1002/ciuz.19970310304.
- ↑ "Johannes Pfleger, Chemist". https://history.evonik.com/sites/geschichte/en/personalities/pfleger-johannes/pages/default.aspx.
- ↑ Elmar Steingruber "Indigo and Indigo Colorants" Ullmann's Encyclopedia of Industrial Chemistry 2004, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a14_149.pub2
External links
 |