Biology:ROP GTPase
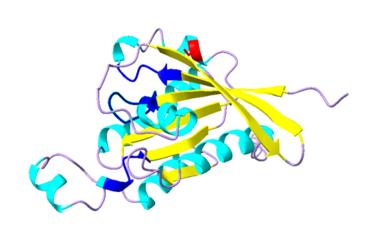
| Dark Blue | possible binding sites at amino acid locations 13-20, 60-64, and 118-121 |
| Yellow | beta sheets |
| Cyan | alpha helices |
| Red | potential phosphorylation site for protein activity regulation; serine residue at amino acid 74 |
| PBD | AF-P92978-F1 |
| UniProt | [1] |
Rho-related GTPases from plants, otherwise known as ROPs, are involved in cell polarity through the regulation of cytoskeleton components like actin and microtubules.[1][2] Unlike mammalian cells, plant cells do not contain heterotrimeric G proteins like Cdc42, Rac, and Rho that are known to regulate cellular polarity.[2]
Structure and Function
ROP proteins are a type of monomeric G proteins found in plants belonging to the Rho family.[1] ROP binding to GTP or GDP determines its activity due to conformational changes within its structure.[1] Within the G-domain of the structure are the G-box motifs G1-5. These motifs are formed during protein folding and are composed of conserved sequences that are responsible for nucleotide and magnesium binding as well as hydrolysis of GTP.[1] Motifs G2 (switch I loop) and G3 (switch II loop) possess distinct conformations depending on GTP binding state.[1] In addition, the G-domain contains a unique and conserved helical domain commonly found in Rho family proteins called αi.[1]
Specific locations within the 3D ROP protein structure, including the amino acids 13-20, 60-64, and 118-121, act as binding sites during protein activity.[3] The serine residue at amino acid 74 has been shown to be a potential protein activity regulation site through phosphorylation.[4]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Ou, Hongxin; Yi, Peishan (13 July 2022). "ROP GTPase-dependent polarity establishment during tip growth in plants". New Phytologist 236 (1): 49–57. doi:10.1111/nph.18373. PMID 35832004. https://dx.doi.org/10.1111/nph.18373.
- ↑ 2.0 2.1 Fu, Ying; Yang, Zhenbiao (1 December 2001). "Rop GTPase: a master switch of cell polarity development in plants". Trends in Plant Science 6 (12): 545–547. doi:10.1016/S1360-1385(01)02130-6. PMID 11738369. https://www.cell.com/trends/plant-science/fulltext/S1360-1385(01)02130-6?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS1360138501021306%3Fshowall%3Dtrue.
- ↑ "UniProt". https://www.uniprot.org/uniprotkb/P92978/entry.
- ↑ Ménesi, Dalma; Klement, Éva; Ferenc, Györgyi; Fehér, Attila (2021). "The Arabidopsis Rho of Plants GTPase ROP1 Is a Potential Calcium-Dependent Protein Kinase (CDPK) Substrate". Plants 10 (10): 2053. 29 September 2021. doi:10.3390/plants10102053. ISSN 2223-7747. PMID 34685862.
 |

