Biology:Animal trypanosomiasis
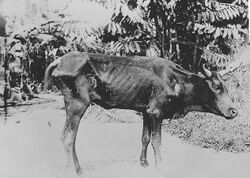
File:Parasite150045-fig1 Dog with Trypanosoma congolense.tif Animal trypanosomiasis, also known as nagana and nagana pest, or sleeping sickness, is a disease of non-human vertebrates. The disease is caused by trypanosomes of several species in the genus Trypanosoma such as T. brucei (which also infects humans to cause African Sleeping Sickness), T. vivax (which causes nagana in livestock mainly in sub-Saharan Africa, although it has also spread to South America[1]), T. congolense and T. simiae.[2][3] The trypanosomes infect the blood of the vertebrate host, causing fever, weakness, and lethargy, which lead to weight loss and anemia. In some animals, the disease is fatal if not treated. In sub-Saharan Africa, trypanosomes are transmitted by tsetse flies,[4] which occur in more than 35 countries.[5]
An interesting feature is the remarkable tolerance to nagana pathology shown by some breeds of cattle, notably the N'Dama – a West African Bos taurus breed. This contrasts with the susceptibility shown by East African B. indicus cattle such as the zebu.[6]
Transmission
Most trypanosomes develop in tsetse flies (Glossina spp.), its biological vector, in about one to a few weeks. When an infected tsetse fly bites an animal, the parasites are transmitted through its saliva. It can also be spread by fomites such as surgical instruments, needles, and syringes. The most important non-Glossina vectors are thought to be horseflies (Tabanidae spp.) and stable flies (Stomoxys spp.).
The immune response of animals may be unable to eliminate trypanosomes completely, and the host may become an inapparent carrier. These inapparent infections can be reactivated if the animal is stressed. Transplacental transmission can also occur.[7]
Transmission was successfully halted on Zanzibar by sterile insect technique (SIT) of the vector Glossina austeni.[8]
Signs and symptoms
The incubation period ranges from four days to approximately eight weeks. The infection leads to significant weight loss and anemia. Various symptoms are observed, including fever, oedema, adenitis, dermatitis and nervous disorders. The disease cannot be diagnosed with certainty except physically detecting parasites by blood microscopic examination or various serological reactions.[7][9]
Vectors
| Disease | Species affected | Trypanosoma agents | Distribution | Glossina vectors |
|---|---|---|---|---|
| Nagana – acute form | antelope
cattle camels horses |
T. brucei brucei | Africa | G. morsitans
G. swynnertoni G. pallidipes G. palpalis G. tachinoides |
| Nagana – chronic form | cattle
camels horses |
T. congolense | Africa | G. palpalis
G. morsitans G. austeni G. swynnertoni G. pallidipes G. longipalpis G. tachinoides G. brevipalpis |
| Nagana – acute form | domestic pigs
cattle camels horses |
T. simiae | Africa | G. palpalis
G. fuscipes G. morsitans G. tachinoides G. longipalpis G. fusca G. tabaniformis G. brevipalpis G. vanhoofi G. austeni |
| Nagana – acute form | cattle
camels horses |
T. vivax | Africa | G. morsitans
G. palpalis G. tachinoides G. swynnertoni G. pallidipes G. austeni G. vanhoofi G. longipalpis |
Control measures
Trypanocidal antiparasitic drugs are the main tool used in Africa to control animal trypanosomiasis. Some drugs can prevent the disease, and are called prophylactic drugs. These can be very effective in protecting animals during the times they are highly exposed to diseases. However, historically these drugs were not always used properly, leading to some resistance.[10][11] Tsetse fly populations can be reduced or eliminated by traps and insecticides. The tsetse habitat can be destroyed by alteration of vegetation, although this approach is no longer deliberately adopted for environmental reasons. In most settings, an integrated, community-based approach to animal trypanosomiasis control is recommended, whereby the control of vectors is combined with the rational use of drugs under the overall guidance and supervision of the national veterinary services.[3][12]
Waterbuck, among other animals, produces chemical odours that repel tsetse flies. This has led to the development of collars that store and gradually release these chemicals, reducing tsetse attack and thus trypanosomiasis incidence for cattle wearing these collars.[13]
Economic impact
Although the loss of direct livestock products (meat, milk, and blood) is problematic, the greatest impact of livestock trypanosomiasis is the loss of crop productivity due to loss of the animals' draught power in the field.[14][15]
Epidemiology
As with other infectious diseases, climate change will have an effect on the distribution and the risk of transmission of animal trypanosomiasis. However, it depends on the geographical region, disease-carrying species, the exact climate change scenario, and many other factors whether there will be a decrease or increase in animal trypanosomiasis.[16]
References
- ↑ Batista, Jael S; Rodrigues, Carla MF; García, Herakles A; Bezerra, Francisco SB; Olinda, Robério G; Teixeira, Marta MG; Soto-Blanco, Benito (May 2011). "Association of Trypanosoma vivax in extracellular sites with central nervous system lesions and changes in cerebrospinal fluid in experimentally infected goats". Veterinary Research 42 (1): 63. doi:10.1186/1297-9716-42-63. PMID 21569364.
- ↑ Diall, Oumar; Cecchi, Giuliano; Wanda, Gift; Argilés-Herrero, Rafael; Vreysen, Marc J. B.; Cattoli, Giovanni; Viljoen, Gerrit J.; Mattioli, Raffaele et al. (2017-07-01). "Developing a Progressive Control Pathway for African Animal Trypanosomosis" (in English). Trends in Parasitology 33 (7): 499–509. doi:10.1016/j.pt.2017.02.005. ISSN 1471-4922. PMID 28456474. https://www.cell.com/trends/parasitology/abstract/S1471-4922(17)30063-6.
- ↑ 3.0 3.1 FAO (2025) (in en). The progressive control pathway for African animal trypanosomosis – Operational guidelines. PAAT Technical and Scientific Series. 13. Rome: FAO. doi:10.4060/cd7991en. ISBN 978-92-5-140405-8. https://openknowledge.fao.org/handle/20.500.14283/cd7991en.
- ↑ "Human African trypanosomiasis (sleeping sickness)". Fact sheet. World Health Organization (WHO). 10 January 2022. https://www.who.int/mediacentre/factsheets/fs259/en/.
- ↑ Cecchi, G.; Paone, M.; de Gier, J.; Zhao, W. (2024) (in English). The continental atlas of the distribution of tsetse flies in Africa. PAAT Technical and Scientific Series, No. 12.. Rome: FAO. doi:10.4060/cd2022en. ISBN 978-92-5-139040-5. https://openknowledge.fao.org/handle/20.500.14283/cd2022en.
- ↑ "Host genetics in African trypanosomiasis". Infection, Genetics and Evolution 8 (3): 229–238. May 2008. doi:10.1016/j.meegid.2008.02.007. PMID 18394971. Bibcode: 2008InfGE...8..229C.
- ↑ 7.0 7.1 "African Animal Trypanosomiasis". October 2018. http://www.cfsph.iastate.edu/Factsheets/pdfs/trypanosomiasis_african.pdf.
- ↑ Caragata, E.; Dong, S.; Dong, Y.; Simoes, M.; Tikhe, C.; Dimopoulos, G. (2020). "Prospects and Pitfalls: Next-Generation Tools to Control Mosquito-Transmitted Disease". Annual Review of Microbiology (Annual Reviews) 74 (1): 455–475. doi:10.1146/annurev-micro-011320-025557. ISSN 0066-4227. PMID 32905752.
- ↑ Finelle, P.. "African animal trypanosomiasis". Food and Agriculture Organization of the United Nations (FAO). http://www.fao.org/docrep/010/ah809e/AH809E02.htm.
- ↑ FAO (2022) (in English). Expert consultation on the sustainable management of parasites in livestock challenged by the global emergence of resistance - Part 2: African animal trypanosomosis and drug resistance – a challenge to progressive, sustainable disease control, 9–10 November 2021.. FAO Animal Production and Health Report No. 18.. Rome: FAO. doi:10.4060/cc2988en. ISBN 978-92-5-137217-3. https://openknowledge.fao.org/handle/20.500.14283/cc2988en.
- ↑ Geerts, S.; Holmes, P.H. (1998). Drug management and parasite resistance in bovine trypanosomiasis in Africa. PAAT Technical and Scientific Series, No. 1.. Rome: FAO. https://openknowledge.fao.org/handle/20.500.14283/w9791en.
- ↑ Bouyer, Jérémy; Bouyer, Fanny; Donadeu, Meritxell; Rowan, Tim; Napier, Grant (2013-11-01). "Community- and farmer-based management of animal African trypanosomosis in cattle" (in English). Trends in Parasitology 29 (11): 519–522. doi:10.1016/j.pt.2013.08.003. ISSN 1471-4922. PMID 24012356. https://www.cell.com/trends/parasitology/abstract/S1471-4922(13)00137-2.
- ↑ Saini, Rajinder K.; Orindi, Benedict O.; Mbahin, Norber; Andoke, John A.; Muasa, Peter N.; Mbuvi, David M.; Muya, Caroline M.; Pickett, John A. et al. (2017-10-17). Solano, Philippe. ed. "Protecting cows in small holder farms in East Africa from tsetse flies by mimicking the odor profile of a non-host bovid" (in en). PLOS Neglected Tropical Diseases 11 (10). doi:10.1371/journal.pntd.0005977. ISSN 1935-2735. PMID 29040267.
- ↑ Swallow, Brent M. (2000). Impacts of trypanosomiasis on African agriculture. PAAT Technical and Scientific Series, No. 2.. Rome: FAO. p. vii+52. ISBN 92-5-104413-9. OCLC 45185699. https://openknowledge.fao.org/handle/20.500.14283/x4755en. AGRIS id XF2001396009. CABD 20003010139.
- ↑ Giordani, Federica; Morrison, Liam J.; Rowan, Timothy G.; De Koning, Harry P.; Barrett, Michael P. (2016-10-10). "The animal trypanosomiases and their chemotherapy: a review". Parasitology (Cambridge University Press (CUP)) 143 (14): 1862–1889. doi:10.1017/s0031182016001268. ISSN 0031-1820. PMID 27719692.
- ↑ Tidman, Rachel; Abela-Ridder, Bernadette; de Castañeda, Rafael Ruiz (2021-01-28). "The impact of climate change on neglected tropical diseases: a systematic review" (in en). Transactions of the Royal Society of Tropical Medicine and Hygiene 115 (2): 147–168. doi:10.1093/trstmh/traa192. ISSN 0035-9203. PMID 33508094. PMC 7842100. https://academic.oup.com/trstmh/article/115/2/147/6121885.
Further reading
- "A step closer to treating nagana". 2017-01-17. http://www.galvmed.org/step-closer-treating-nagana/.
- Cattand, Pierre; de Raadt, Pieter (1991-12-01). "Laboratory Diagnosis of Trypanosomiasis" (in en). Clinics in Laboratory Medicine. Diagnosis of Important Parasitic Diseases 11 (4): 899–908. doi:10.1016/S0272-2712(18)30526-2. ISSN 0272-2712. PMID 1802527.
- Sivajothi, S.; Rayulu, V. C.; Malakondaiah, P.; Sreenivasulu, D. (2016-03-01). "Diagnosis of Trypanosoma evansi in bovines by indirect ELISA" (in en). Journal of Parasitic Diseases 40 (1): 141–144. doi:10.1007/s12639-014-0465-z. ISSN 0975-0703. PMID 27065614.
- Desquesnes, Marc; Gonzatti, Marisa; Sazmand, Alireza; Thévenon, Sophie; Bossard, Géraldine; Boulangé, Alain; Gimonneau, Geoffrey; Truc, Philippe et al. (2022-02-19). "A review on the diagnosis of animal trypanosomoses". Parasites & Vectors 15 (1): 64. doi:10.1186/s13071-022-05190-1. ISSN 1756-3305. PMID 35183235.
- Uilenberg, G. (1998). "African animal trypanosomes". A field guide for the diagnosis, treatment and prevention of African animal trypanosomosis. Food and Agriculture Organization of the United Nations. ISBN 92-5-104238-1. https://www.fao.org/3/X0413E/X0413E02.htm. Retrieved 2023-02-03.
- Mitashi, Patrick; Hasker, Epco; Lejon, Veerle; Kande, Victor; Muyembe, Jean-Jacques; Lutumba, Pascal; Boelaert, Marleen (2012-11-29). "Human African Trypanosomiasis Diagnosis in First-Line Health Services of Endemic Countries, a Systematic Review" (in en). PLOS Neglected Tropical Diseases 6 (11). doi:10.1371/journal.pntd.0001919. ISSN 1935-2735. PMID 23209860.
"Reference Laboratories" (in en-GB). https://www.woah.org/en/what-we-offer/expertise-network/reference-laboratories/.
 |
