Biology:Heterosynaptic plasticity
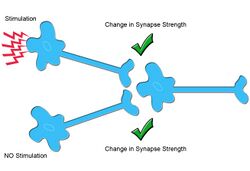
Heterosynaptic plasticity is a subtype of synaptic plasticity, in which changes in synaptic strength are induced by activity at other synapses or by modulatory inputs.[1][2] More broadly, synaptic plasticity refers to molecular changes that are associated with learning and memory, by altering neuronal firing frequency.[3][4] Modern synaptic plasticity theory, building on the work of Santiago Ramón y Cajal, separates plasticity into two categories, Hebbian plasticity, also known as homosynaptic plasticity, and heterosynaptic plasticity. Synaptic plasticity is typically input-specific, meaning that the activity of a postsynaptic neuron, is altered by the release of neurotransmitters from presynaptic neurons in response to stimuli. In homosynaptic plasticity, only the input from a presynaptic neuron synapsing with a specific postsynaptic neuron can undergo plasticity. However, in the case of heterosynaptic plasticity, plasticity can result from either activity in a presynaptic neuron or from a separate modulatory interneuron, resulting in synaptic depression or potentiation. Thus, Hebbian or homosynaptic plasticity is often referred to as activity dependent, while heterosynaptic plasticity is referred to as modulator-dependent. A number of distinct forms of heterosynaptic plasticity have been identified in a variety of brain regions and organisms. These different forms of heterosynaptic plasticity are associated with a variety of neural processes including associative learning, the development of neural circuits, and the homeostasis of synaptic input.[5]
Modulatory input-dependent plasticity (Heterosynaptic Plasticity) Exemplified

Modulatory neurons perform neuromodulation, the release of substances called neuromodulators. Neuromodulators differ from classical neurotransmitters, because they typically do not directly generate electrical responses in target neurons in that they typically do not directly generate electrical responses in target neurons. Rather, the release of neuromodulators can alter the efficacy of neurotransmission in nearby chemical synapses, often producing longer0lasting effects than classical neurotransmitters.[1] The influence of neuromodulators can result in both potentiation and depressive effects, strengthening or weakening synaptic connections on the modulated neuron, regulated by metaplasticity, which refers to changes in plasticity based on the recent history of a specific synapse or cell.[6][2]
A number of neurotransmitters can act as neuromodulators, particularly biogenic amines such as dopamine and serotonin.[5] These neuromodulators can function by activating either ionotropic receptors, ion channels, or G-protein Coupled Receptors (GPCRs), depending on the specific modulator and the type of neuron involved. However, their effects are most commonly mediated through GPCR activation.[2]
The use of these neuromodulators is an example of heterosynaptic plasticity. Released by a neuron called an interneuron, neuromodulators can affect another neuron's communication efficiency with a postsynaptic cell. This is often achieved by altering neurotransmitter release probability, which is stochastic in nature and may be influenced by synaptic vesicle depletion.[1][2] Thus, because the interneuron does not specifically activate the postsynaptic neuron and instead indirectly influences synaptic strength, this mechanism of modulatory input-dependent plasticity is heterosynaptic.[5] Examples of this process include the functions of the neuromodulator serotonin in Aplysia californica and dopamine.
Aplysia californica

A widely used organismal model for studying neuroplasticity and neuronal signaling dynamics is the sea snailAplysia californica.[6] This species has a relatively simple nervous system compared to mammals, facilitating the identification of neural circuits involved in specific behaviors.[1] Studies of plasticity dynamics in Aplysia californica, by Eric Kandel and colleagues, contributed to his receipt of the Nobel Prize in Physiology of Medicine in 2000. Furthermore, these studies provided evidence to the theory that learning and memory are associated with long-term synaptic changes in the brain.[6]
Specifically, heterosynaptic plasticity and neuromdoulation have been demonstrated in early studies. Researchers were able to demonstrate both phenomenons of habituation (similar to Long Term depression or presynaptic depression plasticity), and sensitization (similar to Long Term Potentiation, or presynaptic facilitation plasticity), by examining a gill contraction behavior in response to touch. Even a light touch can serve as simulation of sensory neurons in Aplysia and species containing mechanoreceptors, often referred to as mechanical stimulation.[1][7]
Mechanical stimulation of the siphon (see figure), activated sensory neurons creating enhanced Excitatory Post Synaptic Potentials (EPSP) in the modulatory interneuron. However, repeated stimuli, over time initiated the habituation process, reducing synaptic efficacy, reducing EPSPs on the motor neurons in the gills, ultimately reducing gill response to that stimulus. This process occurs at the sensory-motor neuron synapse that is glutamatergic, and thus it can be said that the glutamatergic synapse is depressed, causing the behavioral habituation (decreased gill response). This is an example of modulatory input-dependent heterosynaptic plasticity, where the sensory neuron functions as a facilitator synapsing with the motor neuron, affecting the motor neuron's firing. Conversely, mechanical stimulation at the tail of Aplysia, generates sensitization of the gill contraction behavior. This stimuli activates sensory neurons to ultimately release serotonin neuromodulators onto the siphon sensory neurons that were previously mentioned, enhancing the release of neurotransmitters from the pre-synaptic cleft of the gill motor neurons, by increasing EPSPs on the siphon sensory neurons.[1] Interestingly, compared to other forms of pre-synaptic plasticity that have effect durations ranging from milliseconds to minutes, this neuromodulatory plasticity is thought to create effects lasting up to one hour.[1][6] Furthermore, when a noxious stimulus was applied to either the head or tail and paired with a light touch to the siphon, it produced a strong motor response, called gill withdrawal reflex. Lastly, evidence for even more long-term plastic change was observed several days later when only a light touch to the siphon elicited the same strong response due to sensitization. These studies serve as evidence for heterosynaptic strengthening between sensory and motor neurons in Aplysia motor circuitry, and as tangible examples of heterosynaptic plasticity.[1][5]
Dopaminergic synapses
Heterosynaptic plasticity is not restricted to serotonin or just a few neuromodulators. In fact, many modulators exist including neuropeptides, cannabinoids, and nitric oxide.[8][9][10] Dopamine in particular has also been shown to act in a neuromodulatory fashion. Much like the serotonin receptors in Aplysia, dopamine receptors are G-protein-coupled receptors that activate cyclic AMP (cAMP) production. This process, however, is important for the storage of memories in mammals, while serotonin's occurs in invertebrates.[5] Within dopaminergic and GABAergic terminals, the neuromodulator dopamine is released from an interneuron altering synapse efficacy on neurons via heterosynaptic plasticity. Commonly, this plasticity can lead to long-term depression, LTD, mediated by dopamine D1 class receptors.[11] The activation of these receptors is required to create LTD and modulate its magnitude, creating decreased synapse efficacy.[12] Further research on dopamine's role in neuromodulation, across different modulators is underway. Experiments performed at the University of Pittsburgh investigated the parallel projects of dopaminergic and GABAergic terminals from the ventral tegmental area to the nucleus accumbens core (NAcCo) in rats, discovering the release of dopamine heterosynaptically triggers LTD at these synapses. Concluding, dopamine is not just a neuromodulator but can also trigger synaptic plasticity independently in neurons.[11] Therefore, heterosynaptic dopamine signaling in mammals can be best represented by dopamine's biological functions of mediating, as well as independently triggering, changes in synaptic plasticity, creating various biological effects.[11]
Heterosynaptic Plasticity's Homeostatic role
Heterosynaptic plasticity may play an important homeostatic role in neural plasticity by normalizing or limiting the total change of synaptic input during ongoing Hebbian plasticity.[13] Hebbian plasticity, an ubiquitous form of homosynaptic, associative plasticity, is believed to underlie learning and memory. Moreover, Hebbian plasticity is induced by and amplifies correlations in neural circuits which creates a positive feedback loop and renders neural circuits unstable. To avoid too much instability Hebbian plasticity needs to be constrained,[14] for instance by the conservation of the total amount of synaptic input. This role is believed to be fulfilled by a diversity of homeostatic mechanisms.
However, to effectively stabilize Hebbian plasticity, which can be induced in a matter of seconds to minutes, homeostatic plasticity has to react rapidly.[15] This requirement, however, is not met by most forms of homeostatic plasticity, which typically act on timescales of hours, days or longer.[16][17] This limitation does not seem to apply to heterosynaptic plasticity.[18][19][20]
To achieve a homeostatic effect, and limit synapse instability, heterosynaptic plasticity serves its homeostatic role through pathway unspecific synaptic changes in the opposite direction of Hebbian plasticity. In other words, whenever homosynaptic long-term potentiation is induced at a given synapse, other unstimulated synapses should be depressed.[21] Conversely, homosynaptic long-term depression would cause other synapses to potentiate in a manner which keeps the average synaptic weight approximately conserved. The scope of these changes could be global or compartmentalized in the dendrites. This theory of balancing the approximate dendritic activity locally, was proposed by Rabinowitch and Segev, and has been further exemplified by hippocampal neurons and neurons in the amygdala.[22]
Developmental Plasticity
The development of neurons, specifically synapses from those almost entirely lacking vesicles pre-synaptically, and forming tight junctions with other neurons, to adult, chemical transmitting capable neurons, is a very long process executed by many biological functions, including contributions by plasticity.[23] Early in development, synaptic connections are not input-specific, most likely because of Ca2+ spillover (i.e. Ca2+ is not restricted to dendrites specifically activated). This spillover represents another mechanism of heterosynaptic change in plasticity. Networks are later refined by input-specific plasticity, which allows for the elimination of connections that are not specifically stimulated.[24] As neuronal circuits mature, it is likely that the concentration of Ca2+ binding proteins increases, which prevents Ca2+ from diffusing to other sites. Increases in localized Ca2+ lead to AMPARs inserted into the membrane. This increase in AMPA density in the postsynaptic membrane increases enables NMDARs to be functional, allowing more Ca2+ to enter the cell.[25] NMDAR subunits also change as neurons mature, increasing the receptor's conductance property.[24][26] These mechanisms facilitate Ca2+ location restriction, and thus specificity, as an organism progresses through development.
In addition to Ca2+ specific mechanisms, increasing total numbers of vesicles in the vesicle pool and extended action potential duration, in young organisms may contribute to observed changes in the same neurons at different ages. For example, studies of the stimulation of MNTB (Medial Nucleus of Trapezoid Body) neurons at a young age has been shown to create a short term depressive effect, however when repeated in mature MNTB neurons, this is not the case. Further possible mechanisms accounting for changes such as in the previous example, may be the alteration of depolarization and hyperpolarization amplitudes throughout development. For example, hyperpolarizing neurons producing Inhibitory Post Synaptic Potentials (IPSPs), became more hyperpolarized throughout development reaching more negative membrane potential values.[23]
Synaptic Scaling
A neural network that undergoes plastic changes between synapses must initiate normalization mechanisms in order to combat unrestrained potentiation or depression. One such mechanism assures that the average firing rate of these neurons is kept at a reasonable rate through synaptic scaling. For example, inhibitory synapses are strengthened or excitatory synapses are weakened to normalize the neural network and allow single neurons to regulate their firing rate, maintaining proper overall synapse activation.[1] Another mechanism is the cell-wide redistribution of synaptic weight. This mechanism conserves the total synaptic weight across the cell by introducing competition between synapses. Thus, normalizing a single neuron after plasticity.[18] During development, cells can be refined when some synapses are preserved and others are discarded to normalize total synaptic weight. In this way, homeostasis is conserved in cells that are undergoing plasticity and normal operation of learning networks is also preserved, allowing new information to be learned.[18] RAI1 influences synaptic scaling indirectly because it regulates gene expression in neurons, including genes involved in synaptic function and neuronal activity. Synaptic scaling depends on proper expression of proteins that adjust synaptic strength up or down to keep neural activity stable. When RAI1 (Retinoic Acid Induced 1) is reduced, this gene regulation is disrupted, which can affect the neuron's ability to maintain that balance and properly carry out homeostatic synaptic scaling [27] NRP2 is involved in pathways that help regulate homeostatic synaptic scaling, which is the process neurons use to keep their activity stable by strengthening or weakening all synapses. When NRP2 (neuropilin-2) activity is reduced, it can lessen signals that promote synaptic downscaling (reducing synaptic strength), allowing synapses to remain stronger and improving neural communication. Decreased NRP2 after exercise may have supported a healthier balance of synaptic activity, making it easier for neurons to maintain stable firing while still adapting and learning.[28]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Purves, D., Augustine, G.J., Fitzpatrick, D., Hall, W.C., LaMantia, A.S., White, L.E. (2012). Synaptic Plasticity. In Neuroscience (5th ed.) (pp. 163-182). Sunderland, Massachusetts: Sinauer Associates.
- ↑ 2.0 2.1 2.2 2.3 Citri, Ami; Malenka, Robert C. (January 2008). "Synaptic Plasticity: Multiple Forms, Functions, and Mechanisms" (in en). Neuropsychopharmacology 33 (1): 18–41. doi:10.1038/sj.npp.1301559. ISSN 1740-634X. PMID 17728696. https://www.nature.com/articles/1301559.
- ↑ Chen, Jen-Yung; Lonjers, Peter; Lee, Christopher; Chistiakova, Marina; Volgushev, Maxim; Bazhenov, Maxim (2013-10-02). "Heterosynaptic Plasticity Prevents Runaway Synaptic Dynamics" (in en). Journal of Neuroscience 33 (40): 15915–15929. doi:10.1523/JNEUROSCI.5088-12.2013. ISSN 0270-6474. PMID 24089497. PMC 3787503. https://www.jneurosci.org/content/33/40/15915.
- ↑ Zenke, Friedemann; Agnes, Everton J.; Gerstner, Wulfram (2015-04-21). "Diverse synaptic plasticity mechanisms orchestrated to form and retrieve memories in spiking neural networks" (in en). Nature Communications 6 (1): 6922. doi:10.1038/ncomms7922. ISSN 2041-1723. PMID 25897632. Bibcode: 2015NatCo...6.6922Z.
- ↑ 5.0 5.1 5.2 5.3 5.4 Bailey, C.H., Giustetto, M., Huang, Y.Y., Hawkins, R.D., Kandel, E.R. (2000) Is heterosynaptic modulation essential for stabilizing Hebbian plasticity and memory. Nature Reviews Neuroscience, 1:1, 11-20.
- ↑ 6.0 6.1 6.2 6.3 Bear, Mark; Connors, Barry; Paradiso, Michael A. (2020). "Molecular Mechanisms of Learning and Memory". Neuroscience: Exploring the Brain, Enhanced Edition (4th ed.). Burlington: Jones & Bartlett Learning, LLC. ISBN 978-1-284-61874-7.
- ↑ Bear, Mark; Connors, Barry; Paradiso, Michael A. (2020). "The Somatic Sensory System". Neuroscience: Exploring the Brain, Enhanced Edition (4th ed.). Burlington: Jones & Bartlett Learning, LLC. ISBN 978-1-284-21128-3.
- ↑ Avery, Michael C.; Krichmar, Jeffrey L. (2017-12-22). "Neuromodulatory Systems and Their Interactions: A Review of Models, Theories, and Experiments" (in English). Frontiers in Neural Circuits 11: 108. doi:10.3389/fncir.2017.00108. ISSN 1662-5110. PMID 29311844.
- ↑ Prast, Helmut; Philippu, Athineos (2001-05-01). "Nitric oxide as modulator of neuronal function". Progress in Neurobiology 64 (1): 51–68. doi:10.1016/S0301-0082(00)00044-7. ISSN 0301-0082. PMID 11250062. https://www.sciencedirect.com/science/article/abs/pii/S0301008200000447.
- ↑ Kendall, Debra A.; Yudowski, Guillermo A. (2017-01-04). "Cannabinoid Receptors in the Central Nervous System: Their Signaling and Roles in Disease" (in English). Frontiers in Cellular Neuroscience 10: 294. doi:10.3389/fncel.2016.00294. ISSN 1662-5102. PMID 28101004.
- ↑ 11.0 11.1 11.2 Ishikawa, M., Otaka, M., Huang, Y. H., Neumann, P. A., Winters, B. D., Grace, A. A., Schlüter, O. M., and Dong, Y. (2013). Dopamine Triggers Heterosynaptic Plasticity. The Journal of Neuroscience, 33(16), 6759-6765.
- ↑ Sajikumar, S., Frey, J. U. (2004). Late-associativity, Synaptic tagging, and the Role of Dopamine during LTP and LTD. Neurobiology of Learning and Memory, 82 (1), 12-25.
- ↑ Royer, S., and Paré, D. (2003). Conservation of total synaptic weight through balanced synaptic depression and potentiation. Nature 422, 518–522.
- ↑ Miller, K.D., and MacKay, D.J. (1994). The role of constraints in Hebbian learning. Neural Comput 6, 100–126.
- ↑ Zenke, F., Hennequin, G., and Gerstner, W. (2013). Synaptic Plasticity in Neural Networks Needs Homeostasis with a Fast Rate Detector. PLoS Comput Biol 9, e1003330.
- ↑ Turrigiano, G.G., and Nelson, S.B. (2004). Homeostatic plasticity in the developing nervous system. Nat Rev Neurosci 5, 97–107.
- ↑ Zenke, F., Gerstner, W., and Ganguli, S. (2017). The temporal paradox of Hebbian learning and homeostatic plasticity. Current Opinion in Neurobiology 43, 166–176.
- ↑ 18.0 18.1 18.2 Chistiakova, M., Volgushev, M. (2009) Heterosynaptic plasticity in the neocortex. Experimental Brain Research, 199, 377-390.
- ↑ Chen, J.-Y., Lonjers, P., Lee, C., Chistiakova, M., Volgushev, M., and Bazhenov, M. (2013). Heterosynaptic Plasticity Prevents Runaway Synaptic Dynamics. J Neurosci 33, 15915–15929.
- ↑ Chistiakova, M., Bannon, N.M., Chen, J.-Y., Bazhenov, M., and Volgushev, M. (2015). Homeostatic role of heterosynaptic plasticity: models and experiments. Front Comput Neurosci 9, 89.
- ↑ Lynch, G.S., Dunwiddie, T., and Gribkoff, V. (1977). Heterosynaptic depression: a postsynaptic correlate of long-term potentiation. Nature 266, 737–739.
- ↑ Pozo, Karine; Goda, Yukiko (2010-05-13). "Unraveling Mechanisms of Homeostatic Synaptic Plasticity" (in English). Neuron 66 (3): 337–351. doi:10.1016/j.neuron.2010.04.028. ISSN 0896-6273. PMID 20471348.
- ↑ 23.0 23.1 Sanes, Dan Harvey; Reh, Thomas A.; Harris, William A. (2005). "Synapse Formation and Function". Development of the nervous system (2nd ed.). Academic Press. pp. 207–247. ISBN 978-0126186215.
- ↑ 24.0 24.1 Sanes, D.H., Reh, T.A., Harris, W.A. (2012). Synapse Formation and Function, Refinement of Synaptic Connections. In Development of the Nervous System (3rd ed.) (pp. 234-274). Boston, Massachusetts: Elsevier Inc.
- ↑ Higley, M.J., Sabatini, B.L. (Feb. 2012.) Calcium Signaling in Dendritic Spines. Cold Spring Harbor Perspectives in Biology. Retrieved from https://cshperspectives.cshlp.org/. doi:10.1101/cshperspect.a005686.
- ↑ Tao, H.W.., Zhang, L.I., Engert, F., Poo, M. (Aug. 2001.) Emergence of Input Specificity of LTP during Development of Retinotectal Connections In Vivo. Neuron: 31, 569-580.
- ↑ Covarelli, J., Vinciarelli, E., Mirarchi, A., Prontera, P., & Arcuri, C. (2025). Retinoic Acid Induced 1 and Smith–Magenis Syndrome: From Genetics to Biology and Possible Therapeutic Strategies. International Journal of Molecular Sciences, 26(14), 6667. https://doi-org.libaccess.sjlibrary.org/10.3390/ijms26146667
- ↑ Savettiere, A., Louras, P., Langdon, S., & Fairchild, J. K. (2025). Identifying Biomarkers of Neuroplasticity Associated with Exercise-Induced Cognitive Change in Older Adults with MCI. Experimental Aging Research, 51(5), 643–656. https://doi-org.libaccess.sjlibrary.org/10.1080/0361073X.2025.2470579
 |
