Biology:Meningitis B
Meningococcal Group B diseases, B-MD, or MenB are meningococcal diseases caused by serotype B of the Neisseria meningitidis bacteria. It is commonly called meningitis B; however not all cases of infection with serotype B cause meningitis. It can also cause meningococcal sepsis. Both are life threatening.[1]
Epidemiology

Serogroup B is the most common cause of meningococcal disease in countries that routinely vaccinate against serotypes A, C, W, and Y.[2] However, overall prevalence of meningococcal disease is low. From 2000 to 2015, only 2 countries had prevalence of B-MD greater than 2/100,000 with most countries reporting incidence of less than 1/100,000.[2][3] The case fatality rate is high, ranging from 10%-20% even after aggressive treatment with antibiotics.[2][3] Three hyper-invasive genotypes are responsible for most outbreaks.[2][3] Asymptomatic carriage of serotype B bacteria in the nasopharyngneal passage is common.[2][3]
MenB vaccines
Vaccines against Meningococcal Group B bacteria are commonly called MenB vaccines.

There are two licensed vaccines specifically against serotype B meningococcal disease widely available; Bexsero (GSK) 4-component MenB vaccine (4CMenB) and Trumenba (Pfizer)[4], a bivalent factor H binding protein vaccine (MenB-FHbp). In the UK and Australia, Bexsero is approved for ages 2 months to 50 years; in the UK and Australia,[5] it is given routinely as part of childhood vaccines as infants the group at greatest risk from meningitis B.[6] The protection offered is short and does not last past early childhood. In the US, both Bexsero and Trumenba are only approved for ages 10-25 and only given to certain high risk groups.[4]
There are additionally two pentavalent vaccines available targeting serogroups A, B, C, W, and Y: Penbraya was approved for use in the United States in October 2023. It combines the vaccines Trumenba and Nimenrix. Penbraya was authorized for medical use in the European Union in November 2024. It is approved for use in individuals 10 through 25 years of age.
Penmenvy was approved for use in the United States in February 2025. Penmenvy is approved for use in people aged 10 through 25 years of age.
Vaccine development and outbreaks
Historically, MenB vaccines were difficult to produce, and required a different approach from vaccines against other serotypes. Whereas effective polysaccharide vaccines have been produced against types A, C, W-135, and Y, the capsular polysaccharide on the type B bacterium is too similar to human neural adhesion molecules to be a useful target.[7]
Several "serogroup B" vaccines have been produced. Strictly speaking, these are not "serogroup B" vaccines, as they do not aim to produce antibodies to the group B antigen: it would be more accurate to describe them as serogroup-independent vaccines, as they employ different antigenic components of the organism; indeed, some of the antigens are common to different Neisseria species.
A vaccine for serogroup B was developed in Cuba in response to a large outbreak of meningitis B during the 1980s. This vaccine was based on artificially produced outer membrane vesicles of the bacterium. The VA-MENGOC-BC vaccine proved safe and effective in randomized double-blind studies,[8][9][10] but it was granted a licence only for research purposes in the United States[11] as political differences limited cooperation between the two countries.[12]
Due to a similarly high prevalence of B-serotype meningitis in Norway between 1974 and 1988, Norwegian health authorities developed a vaccine specifically designed for Norwegian children and adolescents. Clinical trials were discontinued after the vaccine was shown to cover only slightly more than 50% of all cases.[13] Furthermore, lawsuits for damages were filed against the State of Norway by persons affected by serious adverse reactions. Information that the health authorities obtained during the vaccine development was subsequently passed on to Chiron (now GlaxoSmithKline), who later developed a similar vaccine, MeNZB, for New Zealand.[14]
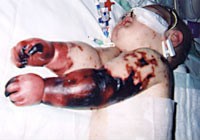
An outbreak in New Zealand beginning in 1991 and lasting until 2006 resulted in 239 deaths, mostly among young children. Of the 5,900 cases, around 20% would suffer some degree of disability.[15] From 2002-2004, the New Zealand Ministry of Health fast-tracked the development of a new vaccine against Meningococcal B, MeNZB.[16] In 2004, the case of Charlotte Cleverley-Bisman became the public face of the disease. The vaccine was approved in 2004 and the vaccination program ran until 2011. The programme was highly successful; in 2006 there were around 300 cases, but by 2010 this had dropped to 30.[15]
A new MenB vaccine was approved for use in Europe in January 2013. Following a positive recommendation from the European Union's Committee for Medicinal Products for Human Use, Bexsero, produced by Novartis, received a licence from the European Commission.[17] However, deployment in individual EU member countries depends on decisions by national governments. As of 2026, Austria, Czechia, Germany, Greece, Hungary, Ireland, Italy, Lithuania, Luxembourg, Malta, Portugal, and Spain routinely vaccinate against MenB.[18]
In July 2013, the United Kingdom's Joint Committee on Vaccination and Immunisation (JCVI) initially issued an interim position statement recommending against the adoption of Bexsero as part of a routine meningococcal B immunisation program, on the grounds of cost-effectiveness.[19] In 2014, the prevalence of B-MD in infants in the UK was approximately 10/100,000 infants.[2][3] The JCVI decision was reverted in favor of Bexsero vaccination in March 2014.[20] In March 2015 the UK government announced that they had reached agreement with GlaxoSmithKline who had taken over Novartis's vaccines business, and that Bexsero would be introduced into the UK routine immunization schedule later in 2015.[21]
In November 2013, in response to an outbreak of B-serotype meningitis on the campus of Princeton University, the acting head of the Centers for Disease Control and Prevention (CDC) meningitis and vaccine-preventable diseases branch told NBC News that they had authorized emergency importation of Bexsero to stop the outbreak.[22] Bexsero was subsequently approved by the FDA in February 2015 for use in individuals 10 through 25 years of age.[23][24] In October 2014, Trumenba, a serogroup B vaccine produced by Pfizer, was approved by the FDA for use in individuals 10 through 25 years of age.[4]
In March 2026, there was an outbreak of meningitis B amongst college and university students in the UK.[25]
References
- ↑ "MenB vaccine for children" (in en). 2024-03-06. https://www.nhs.uk/vaccinations/menb-vaccine-for-children/.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Villena, Rodolfo; Safadi, Marco Aurelio P.; Valenzuela, María Teresa; Torres, Juan P.; Finn, Adam; O'Ryan, Miguel (2018-05-04). "Global epidemiology of serogroup B meningococcal disease and opportunities for prevention with novel recombinant protein vaccines" (in en). Human Vaccines & Immunotherapeutics 14 (5): 1042–1057. doi:10.1080/21645515.2018.1458175. ISSN 2164-5515. PMID 29667483. PMC 5989912. https://www.tandfonline.com/doi/full/10.1080/21645515.2018.1458175.
- ↑ 3.0 3.1 3.2 3.3 3.4 Sridhar, Shruti; Greenwood, Brian; Head, Christopher; Plotkin, Stanley A; Sáfadi, Marco A; Saha, Samir; Taha, Muhamed-Kheir; Tomori, Oyewale et al. (2015-11). "Global incidence of serogroup B invasive meningococcal disease: a systematic review" (in en). The Lancet Infectious Diseases 15 (11): 1334–1346. doi:10.1016/S1473-3099(15)00217-0. https://linkinghub.elsevier.com/retrieve/pii/S1473309915002170.
- ↑ 4.0 4.1 4.2 Research, Center for Biologics Evaluation and (2024-06-20). "TRUMENBA" (in en). FDA. https://www.fda.gov/vaccines-blood-biologics/vaccines/trumenba.
- ↑ https://www.health.gov.au/topics/immunisation/when-to-get-vaccinated/national-immunisation-program-schedule?language=en
- ↑ "Meningitis - Vaccination" (in en). 2018-10-03. https://www.nhs.uk/conditions/meningitis/vaccination/.
- ↑ "An IgG monoclonal antibody to group B meningococci cross-reacts with developmentally regulated polysialic acid units of glycoproteins in neural and extraneural tissues". Journal of Immunology 138 (12): 4402–4407. June 1987. doi:10.4049/jimmunol.138.12.4402. PMID 3108388.
- ↑ "Immune Response Induction and New Effector Mechanisms Possibly Involved in Protection Conferred by the Cuban Anti-Meningococcal BC Vaccine". Infect Immun 69 (7): 4502–8. July 2001. doi:10.1128/IAI.69.7.4502-4508.2001. PMID 11401992.
- ↑ "Outer membrane vesicles of the VA-MENGOC-BC vaccine against serogroup B of Neisseria meningitidis: Analysis of protein components by two-dimensional gel electrophoresis and mass spectrometry". Proteomics 6 (11): 3389–99. June 2006. doi:10.1002/pmic.200500502. PMID 16673438.
- ↑ "Finlay Institute VA-MENGOC-BC Most frequent questions and answers". http://www.finlay.sld.cu/english/products/pregunvamengocbcenglish.htm.
- ↑ "World: Americas Cuba vaccine deal breaks embargo". BBC News Online. 29 July 1999. http://news.bbc.co.uk/2/hi/americas/406780.stm.
- ↑ NBC News Digital (12 November 2015). "Cuban scientist barred from receiving U.S. prize". Associated Press. https://www.nbcnews.com/id/wbna10010619. "A Cuban scientist who helped develop a low-cost synthetic vaccine that prevents meningitis and pneumonia in small children says he was offended the U.S. government denied his request to travel to the United States to receive an award."
- ↑ "Serogroup B meningococcal disease in the Norwegian armed forces: What can we learn from an inconclusive vaccine protection trial?". European Journal of Public Health 7 (3): 261–266. September 1997. doi:10.1093/eurpub/7.3.261. https://academic.oup.com/eurpub/article/7/3/261/503242. Retrieved 28 October 2021.
- ↑ "Investigation: The Meningococcal Gold Rush". Scoop. 7 February 2005. https://www.scoop.co.nz/stories/HL0502/S00064.htm.
- ↑ 15.0 15.1 "New Zealand’s meningococcal B immunisation programme" (in en-GB). 2016-05-30. https://centreforpublicimpact.org/public-impact-fundamentals/new-zealands-meningococcal-b-immunisation-programme/.
- ↑ Lennon, Diana; Jackson, Catherine; Wong, Sharon; Horsfall, Maraekura; Stewart, Joanna; Reid, Stewart (2009-08-15). "Fast tracking the vaccine licensure process to control an epidemic of serogroup B meningococcal disease in New Zealand". Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 49 (4): 597–605. doi:10.1086/603552. ISSN 1537-6591. PMID 19622040. https://web.archive.org/web/20170816191209/https://www.who.int/immunization/sage/6_Diana_Lenon_Fast_Tracking_vaccine_apr_2011.pdf.
- ↑ "First ever MenB vaccine available for use". 24 January 2013. http://www.ovg.ox.ac.uk/OVG/blogs/ojohn/first-ever-menb-vaccine-available-use.
- ↑ "Error: no
|title=specified when using {{Cite web}}". https://vaccine-schedule.ecdc.europa.eu/Scheduler/ByDisease?SelectedDiseaseId=48&SelectedCountryIdByDisease=-1. - ↑ Joint Committee on Vaccination and Immunisation (JCVI) (July 2013). "JCVI interim position statement on use of Bexsero® meningococcal B vaccine in the UK". https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/224896/JCVI_interim_statement_on_meningococcal_B_vaccination_for_web.pdf.
- ↑ "UK review of vaccines flawed, says Novartis' John Porter". March 2014. http://www.pharmatimes.com/Article/14-08-28/UK_review_of_vaccines_flawed_says_Novartis_John_Porter.aspx.
- ↑ "Meningitis B vaccine deal agreed – Jeremy Hunt". BBC News. 29 March 2015. https://www.bbc.co.uk/news/health-32101921.
- ↑ "Emergency meningitis vaccine will be imported to halt Ivy League outbreak". NBC News. 15 November 2013. https://www.nbcnews.com/health/emergency-meningitis-vaccine-will-be-imported-halt-ivy-league-outbreak-2D11603651.
- ↑ "Bexsero". U.S. Food and Drug Administration (FDA). 7 January 2022. https://www.fda.gov/vaccines-blood-biologics/vaccines/bexsero.
- ↑ "FDA approves a second vaccine to prevent serogroup B meningococcal disease" (Press release). U.S. Food and Drug Administration (FDA). 23 January 2015. Archived from the original on 16 March 2015. Retrieved 15 March 2015.
- ↑ UK Health Security Agency (2026-03-18). "Meningitis outbreak: what you need to know" (in en). https://ukhsa.blog.gov.uk/2026/03/18/meningitis-b-outbreak-what-you-need-to-know/.
 |
