Biology:Strategies for Engineered Negligible Senescence
Strategies for Engineered Negligible Senescence (SENS) is the term coined by British biogerontologist Aubrey de Grey for the diverse range of regenerative medical therapies, either planned or currently in development, for the periodical repair of all age-related damage to human tissue with the ultimate purpose of maintaining a state of negligible senescence in the patient, thereby postponing age-associated disease for as long as the therapies are reapplied.[1] The term "negligible senescence" was first used in the early 1990s by professor Caleb Finch to describe organisms such as lobsters and hydras, which do not show symptoms of aging. The term "engineered negligible senescence" first appeared in print in Aubrey de Grey's 1999 book The Mitochondrial Free Radical Theory of Aging.[2] De Grey called SENS a "goal-directed rather than curiosity-driven"[3] approach to the science of aging, and "an effort to expand regenerative medicine into the territory of aging".[4]
While many biogerontologists find it "worthy of discussion",[5][6] some contend that the ultimate goals of de Grey's programme are too speculative given the current state of technology, referring to it as "fantasy rather than science".[7][8]
Framework
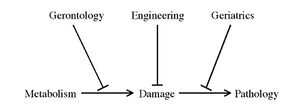
The ultimate objective of SENS is the eventual elimination of age-related diseases and infirmity by repeatedly reducing the state of senescence in the organism. The SENS project consists in implementing a series of periodic medical interventions designed to repair, prevent or render irrelevant all the types of molecular and cellular damage that cause age-related pathology and degeneration, in order to avoid debilitation and death from age-related causes.[1]
The Strategies
The following table as transcribed from the first of de Grey's papers which mentions SENS details the following major ailments and preventative strategies:[9]
| Issue | Countermeasure |
|---|---|
| Accumulation of lysosomal aggregates | Bacterial/fungal hydrolases |
| Accumulation of senescent cells | Senescence marker-targeted toxins |
| Age related tumors | Total telomerase deletion; cell therapy |
| mtDNA mutations | Allotopic mt-coded proteins (e.g. in algae) |
| Immune system damage | IL-7-mediated thymopoiesis |
| AGE buildup | Phenacyldimethylthiazolium chloride |
| Accumulation of extracellular aggregates | Immune-mediated phagocytosis |
| Cell loss | Stem cell therapy, growth factors |
| Hormonal muscle damage | Genetically engineered muscle |
Scientific controversy
While some fields mentioned as branches of SENS are broadly supported by the medical research community, e.g., stem cell research (RepleniSENS), anti-Alzheimers research (AmyloSENS) and oncogenomics (OncoSENS), the SENS programme as a whole has been a highly controversial proposal, with many critics arguing that the SENS agenda is fanciful and the highly complicated biomedical phenomena involved in the aging process contain too many unknowns for SENS to be fully implementable in the foreseeable future.[10] Cancer may well deserve special attention as an aging-associated disease (OncoSENS), but the SENS claim that nuclear DNA damage only matters for aging because of cancer has been challenged in the literature[11] as well as by material in the article DNA damage theory of aging.
In November 2005, 28 biogerontologists published a statement of criticism in EMBO Reports, "Science fact and the SENS agenda: what can we reasonably expect from ageing research?,"[10] arguing "each one of the specific proposals that comprise the SENS agenda is, at our present stage of ignorance, exceptionally optimistic,"[10] and that some of the specific proposals "will take decades of hard work [to be medically integrated], if [they] ever prove to be useful."[10] The researchers argue that while there is "a rationale for thinking that we might eventually learn how to postpone human illnesses to an important degree,"[10] increased basic research, rather than the goal-directed approach of SENS, is presently the scientifically appropriate goal.
More recently, biogerontologist Marios Kyriazis has sharply criticised the clinical applicability of SENS[12][13] claiming that such therapies, even if developed in the laboratory, would be practically unusable by the general public.[14] De Grey responded to one such criticism.[15]
Technology Review controversy
In February 2005, Technology Review, which is owned by the Massachusetts Institute of Technology, published an article by Sherwin Nuland, a Clinical Professor of Surgery at Yale University and the author of "How We Die",[16] that drew a skeptical portrait of SENS, at the time de Grey was a computer associate in the Flybase Facility of the Department of Genetics at the University of Cambridge.
During June 2005, David Gobel, CEO and Co-founder of Methuselah Foundation offered Technology Review $20,000 to fund a prize competition to publicly clarify the viability of the SENS approach. In July 2005, Pontin announced a $20,000 prize, funded 50/50 by Methuselah Foundation and MIT Technology Review, open to any molecular biologist, with a record of publication in biogerontology, who could prove that the alleged benefits of SENS were "so wrong that it is unworthy of learned debate."[17] Technology Review received five submissions to its Challenge. In March 2006, Technology Review announced that it had chosen a panel of judges for the Challenge: Rodney Brooks, Anita Goel, Nathan Myhrvold, Vikram Sheel Kumar, and Craig Venter.[18] Three of the five submissions met the terms of the prize competition. They were published by Technology Review on June 9, 2006. On July 11, 2006, Technology Review published the results of the SENS Challenge.[5]
In the end, no one won the $20,000 prize. The judges felt that no submission met the criterion of the challenge and discredited SENS, although they unanimously agreed that one submission, by Preston Estep and his colleagues, was the most eloquent. Craig Venter succinctly expressed the prevailing opinion: "Estep et al. ... have not demonstrated that SENS is unworthy of discussion, but the proponents of SENS have not made a compelling case for it."[5] Summarizing the judges' deliberations, Pontin wrote that SENS is "highly speculative" and that many of its proposals could not be reproduced with the scientific technology of that period.[clarification needed] Myhrvold described SENS as belonging to a kind of "antechamber of science" where they wait until technology and scientific knowledge advance to the point where it can be tested.[5][6]
SENS Research Foundation
The SENS Research Foundation is a non-profit organization co-founded by Michael Kope, Aubrey de Grey, Jeff Hall, Sarah Marr and Kevin Perrott, which is based in California , United States . Its activities include SENS-based research programs and public relations work for the acceptance of and interest in related research.
See also
References
- ↑ 1.0 1.1 de Grey, Aubrey; Rae, Michael (September 2007). Ending Aging: The Rejuvenation Breakthroughs that Could Reverse Human Aging in Our Lifetime. New York, NY: St. Martin's Press, 416 pp. ISBN:0-312-36706-6.
- ↑ de Grey, Aubrey (November 2003). The Mitochondrial Free Radical Theory of Aging. Austin, Texas: Landes Bioscience. ISBN:1-58706-155-4.
- ↑ Bulkes, Nyssa (March 6, 2006). "Anti-aging research breakthroughs may add up to 25 years to life". The Northern Star. Northern Illinois University (DeKalb, USA).
- ↑ . "Age-Related Diseases: Medicine's Final Adversary?". Huffington Post Healthy Living.
- ↑ 5.0 5.1 5.2 5.3 Pontin, Jason (July 11, 2006). "Is Defeating Aging Only A Dream?". Technology Review.
- ↑ 6.0 6.1 Garreau, Joel (October 31, 2007). "Invincible Man". Washington Post.
- ↑ Warner H et al. (November 2005). "Science fact and the SENS agenda. What can we reasonably expect from ageing research?". EMBO Reports 6 (11): 1006–8. doi:10.1038/sj.embor.7400555. PMID 16264422.
- ↑ Holliday R (April 2009). "The extreme arrogance of anti-aging medicine". Biogerontology 10 (2): 223–8. doi:10.1007/s10522-008-9170-6. PMID 18726707.
- ↑ de Grey, Aubrey D. N. J. (2003-09-01). "The foreseeability of real anti-aging medicine: focusing the debate" (in en). Experimental Gerontology 38 (9): 927–934. doi:10.1016/S0531-5565(03)00155-4. ISSN 0531-5565. http://www.sciencedirect.com/science/article/pii/S0531556503001554.
- ↑ 10.0 10.1 10.2 10.3 10.4 Warner (2005). "Science fact and the SENS agenda. What can we reasonably expect from ageing research?". EMBO Reports 6 (11): 1006–1008. doi:10.1038/sj.embor.7400555. PMID 16264422.
- ↑ Best, BP (2009). "Nuclear DNA damage as a direct cause of aging" (PDF). Rejuvenation Research 12 (3): 199–208. doi:10.1089/rej.2009.0847. PMID 19594328. http://www.benbest.com/lifeext/Nuclear_DNA_in_Aging.pdf.
- ↑ "The impracticality of biomedical rejuvenation therapies: translational and pharmacological barriers". Rejuvenation Research 17 (4): 390–6. 2014. doi:10.1089/rej.2014.1588. PMID 25072550.
- ↑ Kyriazis, Marios (2015). "Translating laboratory anti-aging biotechnology into applied clinical practice: Problems and obstacles". World Journal of Translational Medicine 4 (2): 51–4. doi:10.5528/wjtm.v4.i2.51.
- ↑ "The Fallacy of the Longevity Elixir: Negligible Senescence May be Achieved, but Not by Using Something Physical". Current Aging Science 8 (3): 227–34. 2015. doi:10.2174/1874609808666150702095803. PMID 26135528.
- ↑ "The practicality or otherwise of biomedical rejuvenation therapies: a response to Kyriazis". Rejuvenation Research 17 (4): 397–400. 2014. doi:10.1089/rej.2014.1599. PMID 25072964.
- ↑ Nuland, Sherwin (1994). How We Die: Reflections on Life's Final Chapter. New York: Knopf Random House. ISBN:0-679-41461-4.
- ↑ Pontin, Jason (July 28, 2005). "The SENS Challenge". Technology Review.
- ↑ Pontin, Jason (March 14, 2006). "We've picked the judges for our biogerontology prize". Technology Review.