Chemistry:Debromomarinone
Debromomarinone is a chemical compound isolated from marine actinomycetes.[1]
Biosynthesis
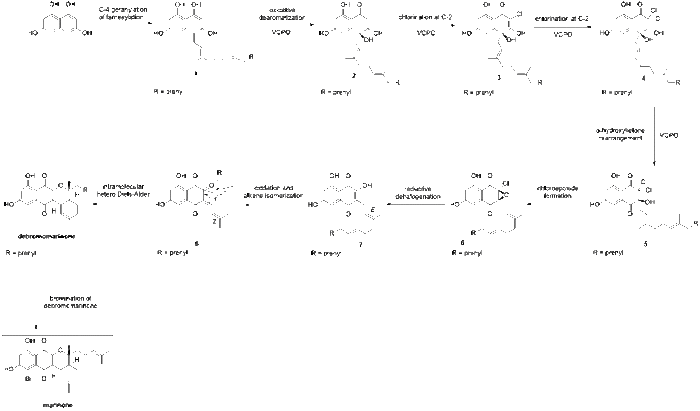
The proposed biosynthesis of marinone was first reported by Murray et al. in 2018.[2] The biosynthesis of marinone begins with 1,3,6,8-tetrahydroxynaphthalene (THN), which is known to be biosynthesized via the condensation of five malonyl-coenzyme A units followed by the aromatization of the resulting pentaketide using a type III polyketide synthase.[3] Next, THN undergoes geranylation or farnesylation at the C-4 position, yielding 1 (Fig. 1). This transformation is catalyzed in vivo by NphB aromatic prenyltransferase in naphterpin biosynthesis[4] or by CnqP3 or CnqP4 in marinone biosynthesis.[5] Then, 1 undergoes oxidative dearomatization which is catalyzed by VCPO, which is a vanadium-dependent chloroperoxidase enzyme. This transformation yields compound 2. Compound 2 then undergoes two consecutive chlorinations at the C2 position, catalyzed by VCPO, to yield 4. Next, a VCPO catalyzed α-hydroxyketone rearrangement shifts the geranyl substituent from C-4 to C-3, yielding 5. Exposure of 5 to mildly basic conditions induces cyclization to yield the α-chloroepoxide, 6. This is followed by the reductive halogenation of the α-chloroepoxide to yield the hydroxynaphthoquinone, 7. Next, oxidation at the C-2 position and facile E/Z isomerization of the double bond affords the enone, 8, which undergoes a intramolecular hetero-Diels-Alder to yield debromomarinone.
References
- ↑ Mikhaĭlov, V. V.; Kuznetsova, T. A.; Eliakov, G. B. (1995). "Bioactive compounds from marine actinomycetes". Bioorganicheskaia Khimiia 21 (1): 3–8. PMID 7710421.
- ↑ 2.0 2.1 "Total Synthesis Establishes the Biosynthetic Pathway to the Naphterpin and Marinone Natural Products". Angewandte Chemie 57 (34): 11009–11014. August 2018. doi:10.1002/anie.201804351. PMID 29935040.
- ↑ "Naphthoquinone-Based Meroterpenoids from Marine-Derived Streptomyces sp. B9173". Biomolecules 10 (8): 1187. August 2020. doi:10.3390/biom10081187. PMID 32824158.
- ↑ "Structural basis for the promiscuous biosynthetic prenylation of aromatic natural products". Nature 435 (7044): 983–987. June 2005. doi:10.1038/nature03668. PMID 15959519. Bibcode: 2005Natur.435..983K.
- ↑ "Studies on terpenoids produced by actinomycetes. 5-dimethylallylindole-3-carboxylic Acid and A80915G-8"-acid produced by marine-derived Streptomyces sp. MS239". The Journal of Antibiotics 61 (2): 75–80. February 2008. doi:10.1038/ja.2008.113. PMID 18408326.
 |
