Chemistry:Norbornadiene
Norbornadiene is an organic compound and a bicyclic hydrocarbon. Norbornadiene is of interest as a metal-binding ligand, whose complexes are useful for homogeneous catalysis. It has been intensively studied owing to its high reactivity and distinctive structural property of being a diene that cannot isomerize (isomers would be anti-Bredt alkenes). Norbornadiene is also a useful dienophile in Diels-Alder reactions.
Synthesis
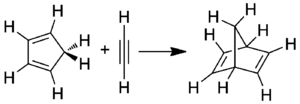
Norbornadiene can be formed by a Diels-Alder reaction between cyclopentadiene and acetylene[1]
Reactions
Quadricyclane, a valence isomer, can be obtained from norbornadiene by a photochemical reaction when assisted by a sensitizer such as acetophenone:[2]
- 300px
The norbornadiene-quadricyclane couple is of potential interest for solar energy storage when controlled release of the strain energy stored in quadricyclane back to norbornadiene is made possible.[3]
Norbornadiene is reactive in cycloaddition reactions. Norbornadiene is also the starting material for the synthesis of diamantane[4] and sumanene and it is used as an acetylene transfer agent for instance in reaction with 3,6-di-2-pyridyl-1,2,4,5-tetrazine.[5]
As a ligand
Norbornadiene is a versatile ligand in organometallic chemistry, where it serves as a two-electron or four-electron donor. (Norbornadiene)molybdenum tetracarbonyl is used as a source of "Mo(CO)4", exploiting the lability of the diene ligand in this case.[6] which is a useful source of "chromium tetracarbonyl," e.g. in reactions with phosphine ligands.
The norbornadiene analogue of cyclooctadiene rhodium chloride dimer has been used in homogeneous catalysis. Chiral, C2-symmetric dienes derived from norbornadiene have also been described.[7][8]
See also
- Norbornane a saturated compound with the same carbon skeleton.
- Norbornene a compound with the same carbon skeleton, but with one less double bond.
References
- ↑ Orrego‐Hernández, Jessica; Hölzel, Helen; Quant, Maria; Wang, Zhihang; Moth‐Poulsen, Kasper (2021-10-14). "Scalable Synthesis of Norbornadienes via in situ Cracking of Dicyclopentadiene Using Continuous Flow Chemistry" (in en). European Journal of Organic Chemistry 2021 (38): 5337–5342. doi:10.1002/ejoc.202100795. ISSN 1434-193X. https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/ejoc.202100795.
- ↑ Smith, Claiborune D. (1988). "Quadricyclane". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=cv6p0962.; Collective Volume, 6, pp. 962
- ↑ Gregory W. Sluggett; Nicholas J. Turro; Heinz D. Roth (1997). "Rh(III)-Photosensitized Interconversion of Norbornadiene and Quadricyclane". J. Phys. Chem. A 101 (47): 8834–8838. doi:10.1021/jp972007h. Bibcode: 1997JPCA..101.8834S.
- ↑ Diamantane in Organic Syntheses Coll. Vol. 6, p.378; Vol. 53, p.30 Online Article
- ↑ Ronald N. Warrener; Peter A. Harrison (2001). "π-Bond Screening in Benzonorbornadienes: The Role of 7-Substituents in Governing the Facial Selectivity for the Diels-Alder Reaction of Benzonorbornadienes with 3,6-Di(2-pyridyl)-s-Tetrazine". Molecules 6 (4): 353–369. doi:10.3390/60400353. PMC 6236418. http://www.mdpi.org/molecules/papers/60400353.pdf.
- ↑ Markus Strotmann; Rudolf Wartchow; Holger Butenschön (2004). "High yield synthesis and structures of some achiral and chiral (diphosphine)tetracarbonylchromium(0) chelate complexes with tetracarbonyl(norbornadiene)chromium(0) as complexation reagent". Arkivoc: KK–1112F. http://www.arkat-usa.org/ark/journal/2004/I13_Krohn/1112/KK-1112F.asp.
- ↑ Ryo Shintani, Tamio Hayashi "Chiral Diene Ligands for Asymmetric Catalysis" Aldrich Chimica Acta 2009, vol. 42, number 2, pp. 31-38.
- ↑ Huang, Yinhua; Hayashi, Tamio (2022). "Chiral Diene Ligands in Asymmetric Catalysis". Chemical Reviews 122 (18): 14346–14404. doi:10.1021/acs.chemrev.2c00218. PMID 35972018.
 |