Chemistry:T cell engager
T-cell engager (TCE) or Bi-specific T-cell engager (BiTE) is a class of artificial bispecific monoclonal antibodies that are investigated for use as anti-cancer drugs. They direct a host's immune system, more specifically the T cells' cytotoxic activity, against cancer cells. BiTE is a registered trademark of Micromet AG (fully owned subsidiary of Amgen Inc).[1]
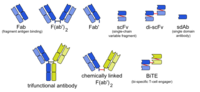
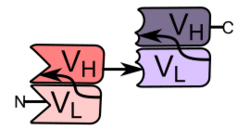
TCE molecules are fusion proteins consisting of two single-chain variable fragments (scFvs) of different antibodies, or amino acid sequences from four different genes, on a single peptide chain of about 55 kilodaltons. One of the scFvs binds to T cells via the CD3 receptor, and the other to a tumor cell via a tumor specific molecule.[2][3]
Mechanism of action

Like other bispecific antibodies, and unlike ordinary monoclonal antibodies, TCEs form a link between T cells and tumor cells. This causes T cells to exert cytotoxic activity on tumor cells by producing proteins like perforin and granzymes, independently of the presence of MHC I or co-stimulatory molecules. These proteins enter tumor cells and initiate the cell's apoptosis.[2][4]
This action mimics physiological processes observed during T cell attacks against tumor cells.[4]
TCEs in clinical assessment or with clinical approvals
Several TCEs are currently in preclinical and clinical trials to assess their therapeutic efficacy and safety.[5]
Blinatumomab
Blinatumomab links T cells with CD19 receptors found on the surface of B cells. The Food and Drug Administration (US) and the European Medicines Agency approved this therapy for adults with Philadelphia chromosome-negative relapsed or refractory acute lymphoblastic leukemia.[6]
Glofitamab
It is a bispecific CD20-directed CD3 T-cell engager. It was approved for medical use in Canada in March 2023, in the United States in June 2023, and in the European Union in July 2023.
Mosunetuzumab
Bispecifically binds CD20 and CD3 to engage T-cells. Mosunetuzumab was approved for medical use in the European Union in June 2022.
Solitomab
Solitomab links T cells with the EpCAM antigen which is expressed by colon, gastric, prostate, ovarian, lung, and pancreatic cancers.[7][8]
Talquetamab
Tarlatamab
Tebentafusp
After clinical trials, in January 2022, the US FDA approved tebentafusp (a BiTE targeting the gp100 peptide) for HLA-A*02:01-positive adult patients with unresectable or metastatic uveal melanoma.[9]
tebentafusp uses the -fusp word stem for fusion proteins as it is not purely made up of antibody pieces. Instead, it is the fusion of three pieces: T cell receptor α chain (synthetic human), T cell receptor β chain (synthetic human), and anti-(human cd3 antigen) immunoglobulin (synthetic scFv fragment). Specifically, it uses the T cell receptor part to bind to a fragment of gp100 presented by HLA-A*02:01 and the ScFv part to bind to CD3.[10]
Epcoritamab
Epcoritamab, sold under the brand names Epkinly & Tepkinly[11][12], is used for the treatment of diffuse large B-cell lymphoma. Epcoritamab is a bispecific CD20-directed CD3 T-cell engager.[11]
Epcoritamab was approved for medical use in the United States in May 2023,[13][14][15][16][17] in the European Union in September 2023, in the UK in October 2023[11] and in Canada in December 2023.
VIR-5500
VIR-5500 is a TCE that targets PSMA and CD3. It incorporates 'PRO-XTEN®' masking to reduce toxicity by confining the activity to the tumor microenvironment, where the mask is cleaved off by tumor-specific proteases.[18] It was in the news during its phase one trial for reducing PSA (a biomarker) in 58 men with metastatic prostate cancer.[19]
Further research
Utilizing the same technology, melanoma (with MCSP specific TCEs) and acute myeloid leukemia (with CD33 specific TCEs) can be targeted.[20] As of 2008[update], research in this area is active.[20]
Another avenue for novel anti-cancer therapies is re-engineering some of the currently used conventional antibodies like trastuzumab (targeting HER2/neu), cetuximab and panitumumab (both targeting the EGF receptor), using the TCE approach.[21]
As of 2009[update], BiTEs against CD66e and EphA2 are being developed as well.[22]
References
- ↑ "US Trademark registration no. 3,068,856, serial number 78/040,636.". US Patent and Trademark Office. http://tarr.uspto.gov/servlet/tarr?regser=serial&entry=78040636.
- ↑ 2.0 2.1 Helwick, Caroline (1 June 2008). "Novel BiTE antibody mediates contact between T cells and cancer cells". Oncology NEWS International 17 (6). http://www.cancernetwork.com/display/article/10165/1165525. Retrieved 15 August 2008.
- ↑ Rüttinger, D.; Zugmaier, G.; Nagorsen, D.; Reinhardt, C.; Baeuerle, P. A. (2008). "BiTE-Antikörper: Durch Bispezifität T-Lymphozyten gegen Tumorzellen richten" (in de). Journal Onkologie (4). http://www.journalonko.de/aktuellview.php?id=1656. Retrieved 9 April 2010.
- ↑ 4.0 4.1 "BiTE Antibody Platform". Micromet Inc. http://www.micromet.de/index.php?id=67.
- ↑ Voynov, V; Adam, PJ (2020). "Discovery Strategies to Maximize the Clinical Potential of T-Cell Engaging Antibodies for the Treatment of Solid Tumors". Antibodies 9 (4): E65–E81. doi:10.3390/antib9040065. PMID 33217946.
- ↑ Malard, Florent; Mohty, Mohamad (4 April 2020). "Acute lymphoblastic leukaemia". The Lancet 395 (10230): 1146–1162. doi:10.1016/s0140-6736(19)33018-1. ISSN 0140-6736. PMID 32247396.
- ↑ Amann, M.; d'Argouges, S.; Lorenczewski, G.; Brischwein, K.; Kischel, R.; Lutterbuese, R.; Mangold, S.; Rau, D. et al. (2009). "Antitumor Activity of an EpCAM/CD3-bispecific BiTE Antibody During Long-term Treatment of Mice in the Absence of T-cell Anergy and Sustained Cytokine Release". Journal of Immunotherapy 32 (5): 452–464. doi:10.1097/CJI.0b013e3181a1c097. PMID 19609237.
- ↑ Kebenko, Maxim; Goebeler, Marie-Elisabeth; Wolf, Martin; Hasenburg, Annette; Seggewiss-Bernhardt, Ruth; Ritter, Barbara; Rautenberg, Beate; Atanackovic, Djordje et al. (2018). "A multicenter phase 1 study of solitomab (MT110, AMG 110), a bispecific EpCAM/CD3 T-cell engager (BiTE®) antibody construct, in patients with refractory solid tumors". Oncoimmunology 7 (8). doi:10.1080/2162402X.2018.1450710. ISSN 2162-4011. PMID 30221040.
- ↑ "FDA approves tebentafusp-tebn for unresectable or metastatic uveal melanoma". 26 January 2022. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-tebentafusp-tebn-unresectable-or-metastatic-uveal-melanoma.
- ↑ "Kimmtrak- tebentafusp injection, solution, concentrate". 26 January 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=24a49f57-d2fc-4ffe-9eb1-fe0460c6b067.
- ↑ 11.0 11.1 11.2 ATC code (2026-01-22). "Summary of Product Characteristics (SmPC)". https://www.medicines.org.uk/emc/product/15188/smpc.
- ↑ "Epcoritamab (Tepkinly®)". 2024-07-01. https://www.macmillan.org.uk/cancer-information-and-support/treatments-and-drugs/epcoritamab.
- ↑ "FDA grants accelerated approval to epcoritamab-bysp for relapsed or refractory diffuse large B-cell lymphoma and high-grade B-cell lymphoma". 19 May 2023. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-grants-accelerated-approval-epcoritamab-bysp-relapsed-or-refractory-diffuse-large-b-cell.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "FDA approves treatment for relapsed or refractory diffuse large B-cell lymphoma and high-grade B-cell lymphoma". 19 May 2023. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-treatment-relapsed-or-refractory-diffuse-large-b-cell-lymphoma-and-high-grade-b-cell.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Epkinly (epcoritamab-bysp) Approved by U.S. FDA as the First and Only Bispecific Antibody to Treat Adult Patients with Relapsed or Refractory Diffuse Large B-Cell Lymphoma (DLBCL)" (Press release). AbbVie. 19 May 2023. Archived from the original on 19 May 2023. Retrieved 20 May 2023 – via PR Newswire.
- ↑ "Drug Approval Package: Epkinly". 26 May 2023. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2023/761324Orig1s000TOC.cfm.
- ↑ "Epcoritamab: First Approval". Drugs 83 (14): 1331–1340. September 2023. doi:10.1007/s40265-023-01930-4. PMID 37597091.
- ↑ "Vir Biotechnology Reports Positive Updated Phase 1 Results for PSMA-targeting, PRO-XTEN® Dual-masked T-Cell Engager VIR-5500 in Patients with Metastatic Prostate Cancer". 2026-01-09. https://investors.vir.bio/news/news-details/2026/Vir-Biotechnology-Reports-Positive-Updated-Phase-1-Results-for-PSMA-targeting-PRO-XTEN-Dual-masked-T-Cell-Engager-VIR-5500-in-Patients-with-Metastatic-Prostate-Cancer/default.aspx.
- ↑ Davis, Nicola (2026-02-28). "Researchers praise 'stunning' results of new prostate cancer treatment". https://www.theguardian.com/society/2026/feb/28/researchers-praise-stunning-results-of-new-prostate-cancer-treatment.
- ↑ 20.0 20.1 Kischel, R (2008). "Characterization in primates of MCSP- and CD33-specific human BiTE antibodies for treatment of Melanoma and AML". Proc Am Assoc Cancer Res 99: Abs 2404. http://www.micromet.de/fileadmin/template/main/pdf/download/AACR_2008_MCSP_CD33_BiTE_Poster.pdf.
- ↑ Lutterbuese, R (2008). "Conversion of cetuximab, panitumumab, trastuzumab and omalizumab into T-cell-engaging BiTE antibodies creates novel drug candidates of high potency". Proc Am Assoc Cancer Res 99: Abs 2402. http://www.micromet.de/fileadmin/template/main/pdf/download/AACR_2008_Herceptin_BiTE_Poster.pdf.
- ↑ Baeuerle, PA; Reinhardt, C (2009). "Bispecific T-cell engaging antibodies for cancer therapy.". Cancer Research 69 (12): 4941–4. doi:10.1158/0008-5472.CAN-09-0547. PMID 19509221.
Further reading
- Kufer, P; Lutterbüse, R; Baeuerle, PA (2004). "A revival of bispecific antibodies". Trends in Biotechnology 22 (5): 238–44. doi:10.1016/j.tibtech.2004.03.006. PMID 15109810.
 |