Earth:Oxygen minimum zone
The oxygen minimum zone (OMZ), sometimes referred to as the shadow zone, is the zone in which oxygen saturation in seawater in the ocean is at its lowest. This zone occurs at depths of about 200 to 1,500 m (700–4,900 ft), depending on local circumstances. OMZs are found worldwide, typically along the western coast of continents, in areas where an interplay of physical and biological processes concurrently lower the oxygen concentration (biological processes) and restrict the water from mixing with surrounding waters (physical processes), creating a "pool" of water where oxygen concentrations fall from the normal range of 4–6 mg/L to below 2 mg/L.[1]
Physical and biological processes
Surface ocean waters generally have oxygen concentrations close to equilibrium with the Earth's atmosphere. In general, colder waters hold more oxygen than warmer waters. As water moves out of the mixed layer into the thermocline, it is exposed to a rain of organic matter from above. Aerobic bacteria feed on this organic matter; oxygen is used as part of the bacterial metabolic process, lowering its concentration within the water. Therefore, the concentration of oxygen in deep water is dependent on the amount of oxygen it had when it was at the surface, minus depletion by deep sea organisms.
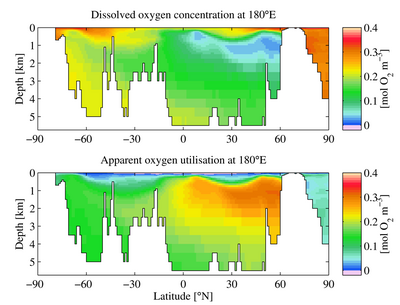
The downward flux of organic matter decreases sharply with depth, with 80–90% being consumed in the top 1,000 m (3,300 ft). The deep ocean thus has higher oxygen because rates of oxygen consumption are low compared with the supply of cold, oxygen-rich deep waters from polar regions. In the surface layers, oxygen is supplied by photosynthesis of phytoplankton. Depths in between, however, have higher rates of oxygen consumption and lower rates of advective supply of oxygen-rich waters. In much of the ocean, mixing processes enable the resupply of oxygen to these waters (see upwelling).
A distribution of the open-ocean oxygen minimum zones is controlled by the large-scale ocean circulation as well as local physical as well as biological processes. For example, wind blowing parallel to the coast causes Ekman transport that upwells nutrients from deep water. The increased nutrients support phytoplankton blooms, zooplankton grazing, and an overall productive food web at the surface. The byproducts of these blooms and the subsequent grazing sink in the form of particulate and dissolved nutrients (from phytodetritus, dead organisms, fecal pellets, excretions, shed shells, scales, and other parts). This "rain" of organic matter (see the biological pump) feeds the microbial loop and may lead to bacterial blooms in water below the euphotic zone due to the influx of nutrients.[3] Since oxygen is not being produced as a byproduct of photosynthesis below the euphotic zone, these microbes use up what oxygen is in the water as they break down the falling organic matter thus creating the lower oxygen conditions.[1]
Physical processes then constrain the mixing and isolate this low oxygen water from outside water. Vertical mixing is constrained due to the separation from the mixed layer by depth. Horizontal mixing is constrained by bathymetry and boundaries formed by interactions with sub-tropical gyres and other major current systems.[4][5][6] Low oxygen water may spread (by advection) from under areas of high productivity up to these physical boundaries to create a stagnant pool of water with no direct connection to the ocean surface even though (as in the Eastern Tropical North Pacific) there may be relatively little organic matter falling from the surface.
Microbes
In OMZs oxygen concentration drops to levels <10 nM at the base of the oxycline and can remain anoxic for over 700 m depth.[7] This lack of oxygen can be reinforced or increased due to physical processes changing oxygen supply such as eddy-driven advection,[7] sluggish ventilation,[8] increases in ocean stratification, and increases in ocean temperature which reduces oxygen solubility.[9]
At a microscopic scale the processes causing ocean deoxygenation rely on microbial aerobic respiration.[9] Aerobic respiration is a metabolic process that microorganisms like bacteria or archaea use to obtain energy by degrading organic matter, consuming oxygen, producing CO2 and obtaining energy in the form of ATP.[9] In the ocean surface photosynthetic microorganisms called phytoplankton use solar energy and CO2 to build organic molecules (organic matter) releasing oxygen in the process.[10] A large fraction of the organic matter from photosynthesis becomes dissolved organic matter (DOM) that is consumed by bacteria during aerobic respiration in sunlit waters. Another fraction of organic matter sinks to the deep ocean forming aggregates called marine snow.[11] These sinking aggregates are consumed via degradation of organic matter and respiration at depth.[8]
At depths in the ocean where no light can reach, aerobic respiration is the dominant process. When the oxygen in a parcel of water is consumed, the oxygen cannot be replaced without the water reaching the surface ocean. When oxygen concentrations drop to below <10 nM, microbial processes that are normally inhibited by oxygen can take place like denitrification and anammox. Both processes extract elemental nitrogen from nitrogen compounds and that elemental nitrogen which does not stay in solution escapes as a gas, resulting in a net loss of nitrogen from the ocean.[8]
Bioavailability of oxygen
Oxygen demand
An organism's demand for oxygen is dependent on its metabolic rate. Metabolic rates can be affected by external factors such as the temperature of the water, and internal factors such as the species, life stage, size, and activity level of the organism. The body temperature of ectotherms (such as fishes and invertebrates) fluctuates with the temperature of the water. As the external temperature increases, ectotherm metabolisms increase as well, increasing their demand for oxygen.[12] Different species have different basal metabolic rates and therefore different oxygen demands.[13][14]
Life stages of organisms also have different metabolic demands. In general, younger stages tend to grow in size and advance in developmental complexity quickly. As the organism reaches maturity, metabolic demands switch from growth and development to maintenance, which requires far fewer resources.[15] Smaller organisms have higher metabolisms per unit of mass, so smaller organisms will require more oxygen per unit mass, while larger organisms generally require more total oxygen.[16] Higher activity levels also require more oxygen.
This is why bioavailability is important in deoxygenated systems: an oxygen quantity which is dangerously low for one species might be more than enough for another species.
Indices and calculations
Several indices to measure bioavailability have been suggested: Respiration Index,[17] Oxygen Supply Index,[18] and the Metabolic Index.[19] The Respiration Index describes oxygen availability based on the free energy available in the reactants and products of the stoichiometric equation for respiration.[17] However, organisms have ways of altering their oxygen intake and carbon dioxide release, so the strict stoichiometric equation is not necessarily accurate.[20] The Oxygen Supply Index accounts for oxygen solubility and partial pressure, along with the Q10 of the organism, but does not account for behavioral or physiological changes in organisms to compensate for reduced oxygen availability.[18] The Metabolic Index accounts for the supply of oxygen in terms of solubility, partial pressure, and diffusivity of oxygen in water, and the organism's metabolic rate.[19] The metabolic index is generally viewed as a closer approximation of oxygen bioavailability than the other indices.
There are two thresholds of oxygen required by organisms:
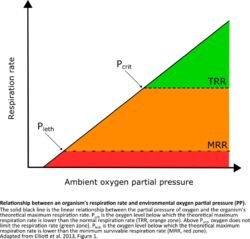
- Pcrit (critical partial pressure)- the oxygen level below which an organism cannot support a normal respiration rate
- Pleth (lethal partial pressure)- the oxygen level below which an organism cannot support the minimum respiration rate necessary for survival.[21][22]
Since bioavailability is specific to each organism and temperature, calculation of these thresholds is done experimentally by measuring activity and respiration rates under different temperature and oxygen conditions, or by collecting data from separate studies.
Life in the OMZ
Despite the low oxygen conditions, organisms have evolved to live in and around OMZs. For those organisms, like the vampire squid, special adaptations are needed to either make do with lesser amounts of oxygen or to extract oxygen from the water more efficiently. For example, the giant red mysid (Gnathophausia ingens) continues to live aerobically (using oxygen) in OMZs. They have highly developed gills with large surface area and thin blood-to-water diffusion distance that enables effective removal of oxygen from the water (up to 90% O2 removal from inhaled water) and an efficient circulatory system with high capacity and high blood concentration of a protein (hemocyanin) that readily binds oxygen.[23][24][25]
Another strategy used by some classes of bacteria in the oxygen minimum zones is to use nitrate rather than oxygen, thus drawing down the concentrations of this important nutrient. This process is called denitrification. The oxygen minimum zones thus play an important role in regulating the productivity and ecological community structure of the global ocean.[26] For example, giant bacterial mats floating in the oxygen minimum zone off the west coast of South America may play a key role in the region's extremely rich fisheries, as bacterial mats the size of Uruguay have been found there.[27]
Zooplankton
Decreased oxygen availability results in decreases in many zooplankton species' egg production, food intake, respiration,[28] and metabolic rates.[29][30][31] Temperature and salinity in areas of decreased oxygen concentrations also affect oxygen availability. Higher temperatures and salinity lower oxygen solubility decrease the partial pressure of oxygen. This decreased partial pressure increases organisms' respiration rates, causing the oxygen demand of the organism to increase.[28][31]
In addition to affecting their vital functions, zooplankton alter their distribution in response to hypoxic or anoxic zones. Many species actively avoid low oxygen zones,[32][33][34] while others take advantage of their predators' low tolerance for hypoxia and use these areas as a refuge.[32][33][34] Zooplankton that exhibit daily vertical migrations to avoid predation and low oxygen conditions also excrete ammonium near the oxycline and contribute to increased anaerobic ammonium oxidation (anammox,[35][31] which produces N2 gas. As hypoxic regions expand vertically and horizontally,[36][37] the habitable ranges for phytoplankton, zooplankton, and nekton increasingly overlap, increasing their susceptibility to predation and human exploitation.[38][29][39][40][33]
Changes
OMZs have changed over time due to effects from numerous global chemical and biological processes.[41] To assess these changes, scientists utilize climate models and sediment samples to understand changes to dissolved oxygen in OMZs.[42] Many recent studies of OMZs have focused on their fluctuations over time and how they may be currently changing as a result of climate change.[42][43]
In geological time scales
Some research has aimed to understand how OMZs have changed over geological time scales.[43] Throughout the history of Earth's oceans, OMZs have fluctuated on long time scales, becoming larger or smaller depending on multiple variables.[44] The factors that change OMZs are the amount of oceanic primary production resulting in increased respiration at greater depths, changes in the oxygen supply due to poor ventilation, and amount of oxygen supplied through thermohaline circulation.[44]
Since industrialization
See also
- Dead zone (ecology), localized areas of dramatically reduced oxygen levels, often due to human impacts.
- Ocean deoxygenation
- Anoxic waters
References
- ↑ 1.0 1.1 Lalli, Carol; Parsons, Timothy (1993). Biological Oceanography: An Introduction. Oxford. ISBN 0-7506-2742-5.
- ↑ "World Ocean Atlas 2009". National Oceanic and Atmospheric Administration. 2009. http://www.nodc.noaa.gov/OC5/WOA09/pr_woa09.html.
- ↑ Mann, K.H.; Lazier, J.R.N. (1991). Dynamics of Marine Ecosystems: Biological-Physical interactions in the oceans.. Blackwell Scientific Publications. ISBN 978-1-4051-1118-8.
- ↑ Gnanadesikan, A.; Bianchi, D.; Pradal, M.A. (2013). "Critical role for mesoscale eddy diffusion in supplying oxygen to hypoxic ocean waters". Geophysical Research Letters 40 (19): 5194–5198. doi:10.1002/grl.50998. Bibcode: 2013GeoRL..40.5194G.
- ↑ Luyten, J; Pedlosky, J; Stommel, H (1983). "The ventilated thermocline". J Phys Oceanogr 13 (2): 292–309. doi:10.1175/1520-0485(1983)013<0292:tvt>2.0.co;2. Bibcode: 1983JPO....13..292L.
- ↑ Pedlosky, J. (1990). "The dynamics of the oceanic subtropical gyres". Science 248 (4953): 316–322. doi:10.1126/science.248.4953.316. PMID 17784484. Bibcode: 1990Sci...248..316P.
- ↑ 7.0 7.1 Bertagnolli, AD; Stewart, FJ (2018). "Microbial niches in marine oxygen minimum zones". Nature Reviews Microbiology 16 (12): 723–729. doi:10.1038/s41579-018-0087-z. PMID 30250271.
- ↑ 8.0 8.1 8.2 Lam, P; Kuypers, MM (2011). "Microbial nitrogen cycling processes in oxygen minimum zones". Annual Review of Marine Science 3: 317–345. doi:10.1146/annurev-marine-120709-142814. PMID 21329208. Bibcode: 2011ARMS....3..317L.
- ↑ 9.0 9.1 9.2 Robinson, C (2019). "Microbial respiration, the engine of ocean deoxygenation". Frontiers in Marine Science 5: 533. doi:10.3389/fmars.2018.00533.
- ↑ Sigman, DM; Hain, MP (2012). "The biological productivity of the ocean". Nature Education Knowledge 3 (6): 1–16.
- ↑ Azam, F; Malfatti, F (2007). "Microbial structuring of marine ecosystems". Nature Reviews Microbiology 5 (10): 782–791. doi:10.1038/nrmicro1747. PMID 17853906.
- ↑ Schulte, PM (2015). "The effects of temperature on aerobic metabolism: towards a mechanistic understanding of the responses of ectotherms to a changing environment". Journal of Experimental Biology 218 (12): 1856–1866. doi:10.1242/jeb.118851. PMID 26085663.
- ↑ Makarieva, AM; Gorshkov, VG; Li, BA; Chown, SL; Reich, PB; Gavrilov, VM (2008). "The effects of temperature on aerobic metabolism: towards a mechanistic understanding of the responses of ectotherms to a changing environment". Proceedings of the National Academy of Sciences 105 (44): 16994–16999. doi:10.1073/pnas.0802148105. PMID 18952839.
- ↑ Balmer, RT (2011). Modern Engineering Dynamics. Academic Press.
- ↑ Rosenfeld, J; Van Leeuwen, T; Richards, J; Allen, D (2015). "Relationship between growth and standard metabolic rate: measurement artefacts and implications for habitat use and life-history adaptation in salmonids". Journal of Animal Ecology 84 (1): 4–20. doi:10.1111/1365-2656.12260. PMID 24930825. Bibcode: 2015JAnEc..84....4R.
- ↑ Singer, D (2004). "Metabolic adaptation to hypoxia: cost and benefit of being small". Respiratory Physiology & Neurobiology 141 (3): 215–228. doi:10.1016/j.resp.2004.02.009. PMID 15288595.
- ↑ 17.0 17.1 Brewer, PG; Peltzer, ET (2009). "Limits to Marine Life". Science 324 (5925): 347–348. doi:10.1126/science.1170756. PMID 19372421.
- ↑ 18.0 18.1 Verberk, WCEP; Bilton, DT; Calosi, P; Spicer, JI (2011). "Oxygen supply in aquatic ectotherms: partial pressure and solubility together explain biodiversity and size patterns.". Ecology 92 (8): 1565–1572. doi:10.1890/10-2369.1. PMID 21905423. Bibcode: 2011Ecol...92.1565V.
- ↑ 19.0 19.1 Deutsch, C; Ferrel, A; Seibel, B; Pörtner, HO; Huey, R (2015). "Climate change tightens a metabolic constraint on marine habitats". Science 348 (6239): 1132–1135. doi:10.1126/science.aaa1605. PMID 26045435. Bibcode: 2015Sci...348.1132D.
- ↑ Seibel, BA; Childress, JJ (2013). "The real limits to marine life: a further critique of the Respiration Index". Biogeosciences 10 (5): 2815. doi:10.5194/bg-10-2815-2013. Bibcode: 2013BGeo...10.2815S.
- ↑ Pörtner, HO (2010). "Oxygen- And capacity-limitation of thermal tolerance: A matrix for integrating climate-related stressor effects in marine ecosystems". Journal of Experimental Biology 213 (6): 881–893. doi:10.1242/jeb.037523. PMID 20190113.
- ↑ Elliott, DT; Pierson, JJ; Roman, MR (2013). "Elliott, D.T., Pierson, J.J. and Roman, M.R., 2013. Predicting the effects of coastal hypoxia on vital rates of the planktonic copepod Acartia tonsa Dana". PLOS ONE 8 (5). doi:10.1371/journal.pone.0063987. PMID 23691134.
- ↑ Childress, J.J.; Seibel, B.A. (1998). "Life at stable low oxygen levels: adaptations of animals to oceanic oxygen minimum layers.". The Journal of Experimental Biology 201 (Pt 8): 1223–1232. doi:10.1242/jeb.201.8.1223. PMID 9510533. https://digitalcommons.uri.edu/bio_facpubs/15.
- ↑ Sanders, N.K.; Childress, J.J. (1990). "Adaptations to the Deep-Sea Oxygen Minimum Layer: Oxygen Binding by the Hemocyanin of the Bathypelagic Mysid, Gnathophausia ingens Dohrn". Biological Bulletin 178 (3): 286–294. doi:10.2307/1541830. PMID 29314949. https://www.biodiversitylibrary.org/part/7475.
- ↑ Torres, J.J.; Grigsby, M.D.; Clarke, M.E. (2012). "Aerobic and anaerobic metabolism in oxygen minimum layer fishes: the role of alcohol dehydrogenase". The Journal of Experimental Biology 215 (11): 1905–1914. doi:10.1242/jeb.060236. PMID 22573769.
- ↑ Deutsch, Curtis; Sarmiento, Jorge L.; Sigman, Daniel M.; Gruber, Nicolas; Dunne, John P. (2006). "Spatial coupling of nitrogen inputs and losses in the ocean". Nature 445 (7124): 163–7. doi:10.1038/nature05392. PMID 17215838. Bibcode: 2007Natur.445..163D.
- ↑ Leahy, Stephen (20 April 2010). "Giant Bacteria Colonise the Oceans". Tierramérica. Inter Press Service. http://www.ipsnews.net/news.asp?idnews=51117.
- ↑ 28.0 28.1 Elliott, DT; Pierson, JJ; Roman, MR (2013). "Elliott, D.T., Pierson, J.J. and Roman, M.R., 2013. Predicting the effects of coastal hypoxia on vital rates of the planktonic copepod Acartia tonsa Dana". PLOS ONE 8 (5). doi:10.1371/journal.pone.0063987. PMID 23691134.
- ↑ 29.0 29.1 Elder, LE; Seibel, BA (2015). "Ecophysiological implications of vertical migration into oxygen minimum zones for the hyperiid amphipod Phronima sedentaria". Journal of Plankton Research 37 (5): 897–911. doi:10.1093/plankt/fbv066.
- ↑ Seibel, BA (2011). "Critical oxygen levels and metabolic suppression in oceanic oxygen minimum zones". Journal of Experimental Biology 214 (2): 326–336. doi:10.1242/jeb.049171. PMID 21177952.
- ↑ 31.0 31.1 31.2 Kiko, R; Hauss, H; Bucholz, F; Melzner, F (2016). "Ammonium excretion and oxygen respiration of tropical copepods and euphausiids exposed to oxygen minimum zone conditions". Biogeosciences 13 (8): 2241–2255. doi:10.5194/bg-13-2241-2016. Bibcode: 2016BGeo...13.2241K.
- ↑ 32.0 32.1 Elliott, DT; Pierson, JJ; Roman, MR (2012). "Relationship between environmental conditions and zooplankton community structure during summer hypoxia in the northern Gulf of Mexico.". Journal of Plankton Research 34 (7): 602–613. doi:10.1093/plankt/fbs029.
- ↑ 33.0 33.1 33.2 Vanderploeg, HA; Ludsin, SA; Cavaletto, JF; Höök, TO; Pothoven, SA; Brandt, SB; Liebig, JR; Lang, GA (2009). "Hypoxic zones as habitat for zooplankton in Lake Erie: refuges from predation or exclusion zones?". Journal of Experimental Marine Biology and Ecology 381: S108–S120. doi:10.1016/j.jembe.2009.07.015. Bibcode: 2009JEMBE.381S.108V.
- ↑ 34.0 34.1 Vanderploeg, HA; Ludsin, SA; Ruberg, SA; Höök, TO; Pothoven, SA; Brandt, SB; Lang, GA; Liebig, JR et al. (2009). "Hypoxia affects spatial distributions and overlap of pelagic fish, zooplankton, and phytoplankton in Lake Erie". Journal of Experimental Marine Biology and Ecology 381: S92–S107. doi:10.1016/j.jembe.2009.07.027. Bibcode: 2009JEMBE.381S..92V.
- ↑ Bianchi, D; Babbin, AR; Galbraith, ED (2014). "Enhancement of anammox by the excretion of diel vertical migrators". Proceedings of the National Academy of Sciences 111 (44): 15653–15658. doi:10.1073/pnas.1410790111. PMID 25288743. Bibcode: 2014PNAS..11115653B.
- ↑ Stramma, L; Prince, ED; Schmidtko, S; Luo, J; Hoolihan, JP; Visbeck, M; Wallace, DWR; Brandt, P et al. (2012). "Expansion of oxygen minimum zones may reduce available habitat for tropical pelagic fishes". Nature Climate Change 2 (1): 33–37. doi:10.1038/nclimate1304. Bibcode: 2012NatCC...2...33S. http://oceanrep.geomar.de/13127/2/Stramma.pdf.
- ↑ Prince, ED; Goodyear, CP (2006). "Hypoxia-based habitat compression of tropical pelagic fishes". Fisheries Oceanography 15 (6): 451–464. doi:10.1111/j.1365-2419.2005.00393.x. Bibcode: 2006FisOc..15..451P.
- ↑ de Mutsert, K; Steenbeek, J; Lewis, K; Buszowski, J; Cowan Jr., JH; Christensen, V (2016). "Exploring effects of hypoxia on fish and fisheries in the northern Gulf of Mexico using a dynamic spatially explicit ecosystem model". Ecological Modelling 331: 142–150. doi:10.1016/j.ecolmodel.2015.10.013. Bibcode: 2016AGUOSAH43A..07D.
- ↑ Kraus, RT; Secor, DH; Wingate, RL (2015). "Testing the thermal-niche oxygen-squeeze hypothesis for estuarine striped bass". Environmental Biology of Fishes 98 (10): 2083–2092. doi:10.1007/s10641-015-0431-3. Bibcode: 2015EnvBF..98.2083K.
- ↑ Roman, MR; Pierson, JJ; Kimmel, DG; Boicourt, WC; Zhang, X (2012). "Impacts of hypoxia on zooplankton spatial distributions in the northern Gulf of Mexico". Estuaries and Coasts 35 (5): 1261–1269. doi:10.1007/s12237-012-9531-x. Bibcode: 2012EstCo..35.1261R.
- ↑ US Department of Commerce, National Oceanic and Atmospheric Administration. "What is a dead zone?" (in EN-US). https://oceanservice.noaa.gov/facts/deadzone.html.
- ↑ 42.0 42.1 Stramma, Lothar; Schmidtko, Sunke; Levin, Lisa A.; Johnson, Gregory C. (April 2010). "Ocean oxygen minima expansions and their biological impacts". Deep Sea Research Part I: Oceanographic Research Papers 57 (4): 587–595. doi:10.1016/j.dsr.2010.01.005. ISSN 0967-0637. Bibcode: 2010DSRI...57..587S.
- ↑ 43.0 43.1 van Geen, A.; Smethie, W. M.; Horneman, A.; Lee, H. (2006-10-07). "Sensitivity of the North Pacific oxygen minimum zone to changes in ocean circulation: A simple model calibrated by chlorofluorocarbons". Journal of Geophysical Research 111 (C10): C10004. doi:10.1029/2005jc003192. ISSN 0148-0227. Bibcode: 2006JGRC..11110004V.
- ↑ 44.0 44.1 Cartapanis, Olivier; Tachikawa, Kazuyo; Bard, Edouard (2011-10-29). "Northeastern Pacific oxygen minimum zone variability over the past 70 kyr: Impact of biological production and oceanic ventilation". Paleoceanography 26 (4): PA4208. doi:10.1029/2011pa002126. ISSN 0883-8305. Bibcode: 2011PalOc..26.4208C.
 |
