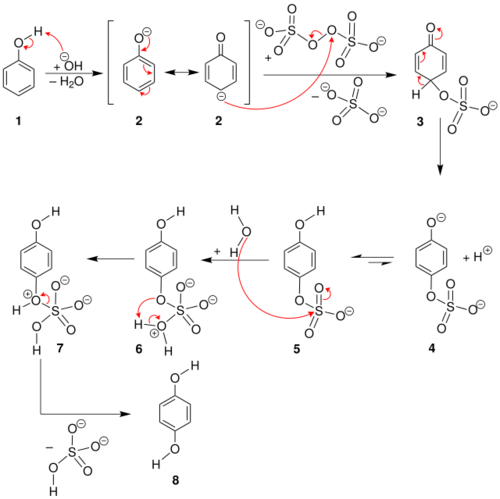Chemistry:Elbs persulfate oxidation
| Elbs persulfate oxidation | |
|---|---|
| Named after | Karl Elbs |
| Reaction type | Organic redox reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000179 |
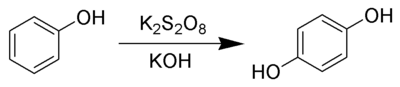
The Elbs persulfate oxidation is the organic reaction of phenols with alkaline potassium persulfate to form para-diphenols.[1] The reaction is generally performed in water at room temperatures or below, using equimolar quantities of reagents.
Several reviews have been published.[2][3][4]
Scope and mechanism
The reaction is disadvantaged by moderate to low chemical yields with recovery of starting material and complete consumption of the persulfate.[4] It is suggested that the phenol in many cases is a catalyst converting the persulfate into a sulfate. Despite this, the Elbs reaction remains generally useful in a research setting, as it is simple to perform and is tolerant of a wide range of other functional groups, which are not oxidised under these conditions.[4]
A reaction mechanism has been postulated which accounts for the observed para substitution featuring the tautomeric para carbanion of the starting phenolate ion:[5] It begins with nucleophilic displacement on the peroxide oxygen of the peroxodisulfate (peroxydisulfate) ion, to give an intermediate sulfate group (3), which is then hydrolyzed to the hydroxyl group.
See also
- Boyland–Sims oxidation
- Dakin reaction
References
- ↑ Elbs, K. (1893). "Ueber Nitrohydrochinon" (in German). J. Prakt. Chem. 48: 179–185. doi:10.1002/prac.18930480123. https://zenodo.org/record/1427970.
- ↑ Sethna, S. M. (1951). "The Elbs Persulfate Oxidation". Chem. Rev. 49 (1): 91–101. doi:10.1021/cr60152a002.
- ↑ Lee, J. B.; Uff, B. C. (1967). "Organic reactions involving electrophilic oxygen". Quart. Rev. 21 (4): 453. doi:10.1039/qr9672100429.
- ↑ 4.0 4.1 4.2 Behrman, E. J. (1988). "The Persulfate Oxidation of Phenols and Arylamines (The Elbs and the Boyland-Sims Oxidations)". Org. React.. 35. pp. 421–511. doi:10.1002/0471264180.or035.02. ISBN 0471264180.
- ↑ Behrman, E. J. (2006). "The Elbs and Boyland-Sims peroxydisulfate oxidations". Beilstein Journal of Organic Chemistry 2 (1): 22. doi:10.1186/1860-5397-2-22. PMID 17090305.
Add the following: E. J. Behrman, The Elbs & Boyland-Sims Oxidations: An Updated Literature Survey. Mini-Rev. Org. Chem, 18(2021)621-625.
 |