Biology:CVNH domain
| CVNH | |||||||||
|---|---|---|---|---|---|---|---|---|---|
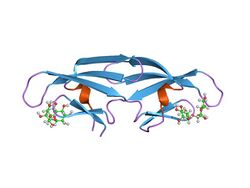 solution nmr structure of complex of 1:2 cyanovirin-n:man-alpha1,2-man-alpha restrained regularized mean coordinates | |||||||||
| Identifiers | |||||||||
| Symbol | CVNH | ||||||||
| Pfam | PF08881 | ||||||||
| InterPro | IPR011058 | ||||||||
| |||||||||
In molecular biology, the CVNH domain (CyanoVirin-N Homology domain) is a conserved protein domain. It is found in the sugar-binding antiviral protein cyanovirin-N (CVN) as well as proteins from filamentous ascomycetes and in the fern Ceratopteris richardii.[1]
Cyanovirin-N (CV-N) is an 11-kDa protein from the cyanobacterium Nostoc ellipsosporum that displays virucidal activity against several viruses, including human immunodeficiency virus (AIDS). The virucidal activity of CV-N is mediated through specific high-affinity interactions with the viral surface envelope glycoproteins gp120 and gp41, as well as to high-mannose oligosaccharides found on the HIV envelope.[2] In addition, CV-N is active against rhinoviruses, human parainfluenza virus, respiratory syncytial virus, and enteric viruses. The virucidal activity of CV-N against influenza virus is directed towards viral haemagglutinin.[3] CV-N has a complex fold composed of a duplication of a tandem repeat of two homologous motifs comprising three-stranded beta sheet and beta hairpins.[4]
References
- ↑ "The anti-HIV cyanovirin-N domain is evolutionarily conserved and occurs as a protein module in eukaryotes". Proteins 60 (4): 670–8. September 2005. doi:10.1002/prot.20543. PMID 16003744.
- ↑ "Cyanovirin-N: a sugar-binding antiviral protein with a new twist". Cell. Mol. Life Sci. 60 (2): 277–287. 2003. doi:10.1007/s000180300023. PMID 12678493.
- ↑ "Potent anti-influenza activity of cyanovirin-N and interactions with viral hemagglutinin". Antimicrob. Agents Chemother. 47 (8): 2518–2525. 2003. doi:10.1128/aac.47.8.2518-2525.2003. PMID 12878514.
- ↑ "Structures of the complexes of a potent anti-HIV protein cyanovirin-N and high mannose oligosaccharides". J. Biol. Chem. 277 (37): 34336–34342. 2002. doi:10.1074/jbc.M205909200. PMID 12110688.
 |
