Chemistry:Achmatowicz reaction
| Achmatowicz reaction | |
|---|---|
| Named after | Osman Achmatowicz Jr. |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000233 |
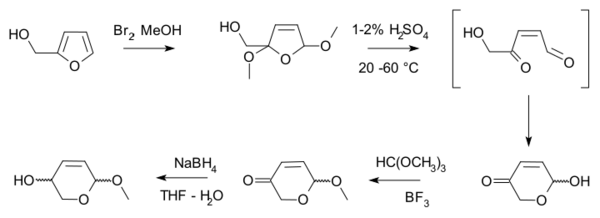
The Achmatowicz reaction, also known as the Achmatowicz rearrangement or the Achmatowicz oxidation[1], is an organic synthesis in which a furan is converted to a dihydropyran. In the original publication by the Polish chemist Osman Achmatowicz Jr. (b. 20 December 1931 in Vilnius) in 1971 [2] furfuryl alcohol is reacted with bromine in methanol to 2,5-dimethoxy-2,5-dihydrofuran which rearranges to the dihydropyran with dilute sulfuric acid. Additional reaction steps, alcohol protection with methyl orthoformate and boron trifluoride) and then ketone reduction with sodium borohydride produce an intermediate from which many monosaccharides can be synthesised.
The Achmatowicz protocol has been used in total synthesis, including those of desoxoprosophylline,[3] pyrenophorin [4][5] Recently it has been used in diversity oriented synthesis[6][7]
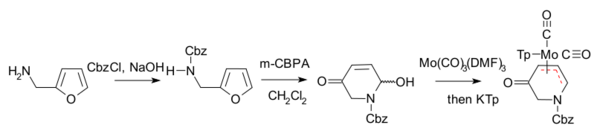
and in enantiomeric scaffolding.[8][9]
References
- ↑ Taber, Douglass F. (4 Feb 2019). "The He/Lee synthesis of basiliolide C". https://www.organic-chemistry.org/Highlights/2019/04February.shtm.
- ↑ Achmatowicz, O.; Bukowski, P.; Szechner, B.; Zwierzchowska, Z.; Zamojski, A. (1971-01-01). "Synthesis of methyl 2,3-dideoxy-DL-alk-2-enopyranosides from furan compounds: A general approach to the total synthesis of monosaccharides". Tetrahedron 27 (10): 1973–1996. doi:10.1016/S0040-4020(01)98229-8. ISSN 0040-4020.
- ↑ Yang, Cui-Fen; Xu, Yi-Ming; Liao, Li-Xin; Zhou, Wei-Shan (1998-12-10). "Asymmetric total synthesis of (+)-desoxoprosophylline". Tetrahedron Letters 39 (50): 9227–9228. doi:10.1016/S0040-4039(98)02129-7. ISSN 0040-4039.
- ↑ Kobayashi, Yuichi; Nakano, Miwa; Kumar, G. Biju; Kishihara, Kiyonobu (1998-10-01). "Efficient Conditions for Conversion of 2-Substituted Furans into 4-Oxygenated 2-Enoic Acids and Its Application to Synthesis of (+)-Aspicilin, (+)-Patulolide A, and (−)-Pyrenophorin". The Journal of Organic Chemistry 63 (21): 7505–7515. doi:10.1021/jo980942a. ISSN 0022-3263. PMID 11672404.
- ↑ Zhang, Yongqiang; Liebeskind, Lanny S. (2006-01-01). "Organometallic Enantiomeric Scaffolding: Organometallic Chirons. Total Synthesis of (−)-Bao Gong Teng A by a Molybdenum-Mediated [5+2] Cycloaddition". Journal of the American Chemical Society 128 (2): 465–472. doi:10.1021/ja055623x. ISSN 0002-7863. PMID 16402833. Bibcode: 2006JAChS.128..465Z.
- ↑ Reagents: NBS, PPTS, for macrobeads see: solid-phase synthesis
- ↑ Burke, Martin D.; Berger, Eric M.; Schreiber, Stuart L. (2004-11-01). "A Synthesis Strategy Yielding Skeletally Diverse Small Molecules Combinatorially". Journal of the American Chemical Society 126 (43): 14095–14104. doi:10.1021/ja0457415. ISSN 0002-7863. PMID 15506774. Bibcode: 2004JAChS.12614095B.
- ↑ Coombs, Thomas C.; Lee; Wong, Heilam; Armstrong, Matthew; Cheng, Bo; Chen, Wenyong; Moretto, Alessandro F.; Liebeskind, Lanny S. (2008-02-01). "Practical, Scalable, High-Throughput Approaches to η3-Pyranyl and η3-Pyridinyl Organometallic Enantiomeric Scaffolds Using the Achmatowicz Reaction". The Journal of Organic Chemistry 73 (3): 882–888. doi:10.1021/jo702006z. ISSN 0022-3263. PMID 18171075.
- ↑ Reagents: benzyl chloroformate protects amine as Cbz group, Achmatowitz reaction with m-CPBA, complexation with a molybdenum compound, Cp is cyclopentadienyl anion
 |