Chemistry:Arsonic acid
From HandWiki
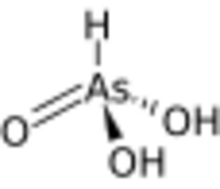
| |
| Names | |
|---|---|
| Preferred IUPAC name
Arsonic acid[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| AsH3O3 | |
| Molar mass | 125.943 g·mol−1 |
| Conjugate base | Hydrogen arsonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
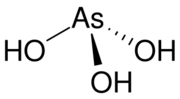

Arsonic acid is the simplest of the arsonic acids. It is a hypothetical compound, although the tautomeric arsenious acid (As(OH)3) is well established. In contrast to the instability of HAsO(OH)2, the phosphorus compound with analogous stoichiometry exists as the tetrahedral tautomer. Similarly, organic derivatives such as phenylarsonic acid are tetrahedral with pentavalent central atom.[3]
There are similar acids that are the same except for having different pnictogens. The phosphorus equivalent is phosphonic acid.
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 916. doi:10.1039/9781849733069-00648. ISBN 978-0-85404-182-4.
- ↑ Munoz-Hernandez, M.-A. (1994). "Arsenic: Inorganic Chemistry". in King, R. B.. Encyclopedia of Inorganic Chemistry. Chichester: John Wiley & Sons.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
 |
