Chemistry:Atrane
Atranes are a class of tricyclic molecules that are fused to a common central bond. Unlike the propellanes, the central bond is a transannular dative bond from a Lewis base—typically nitrogen—at one bridgehead to a Lewis acidic atom such as silicon or boron at the other bridgehead.[1] Each of the three rings is therefore heterocyclic. Each of the rings is typically five atoms, or sometimes six atoms.[2] The name "atrane" was first proposed by Mikhail Grigorievich Voronkov (ru).[1]
Nomenclature
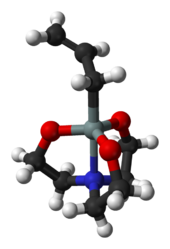

Various atranes are named depending on the central element, e.g. "silatrane" (E = silicon); "boratrane" (E = boron); "phosphatrane" (E = phosphorus), stannatrane (E = tin), etc. It is also proposed that when Y = nitrogen, the prefix "aza" be inserted before element + "atrane" (azasilatrane, for example) because atranes wherein E = silicon and Y = oxygen have been referred to as just "silatranes".[4]
Bis-atranes are two atrane cages that are fused end-to-end, sharing the same Lewis-acid donor atom in two co-axial dative interations.[5]
Structure and properties
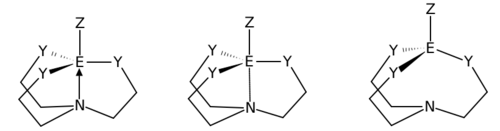
Silatranes exhibit unusual properties, as the transannular coordinate bond in atranes can be stretched (quasiatranes) and even broken (proatranes). The strength (and multiplicity) of the central bond depends on the stereoelectronic properties of the surrounding ligands, the electronegativity of the participating atoms, and the size of the rings. A strong driving force for the formation of the central bond is relief of ring strain from the otherwise-formed 8-membered rings.[4]
Atranes exhibit biological activity in which the coordination of nitrogen to silane plays an important role. Some derivatives such as phenylsilatrane are highly toxic.

Proazaphosphatrane is a very strong non-ionic base and is utilized in various types of organic synthesis as an efficient catalyst.
See also
References
- ↑ 1.0 1.1 Voronkov, Mikhail G.; Baryshok, Viktor P. "Atranes - a new generation of biologically active substances" (in Russian) Vestnik Rossiiskoi Akademii Nauk 2010, volume 80, 985-992.
- ↑ Kim, Da Jung; Hong, Younjin; Kim, So Han; Lee, Kang Mun; Mun, Sang-Deok; Yoon, Sungwoo; Lee, Junseong; Do, Youngkyu et al. (2011). "Boratranes with all six-membered rings or with two different ring sizes: Synthesis, characterization, and X-ray crystal structures". Inorganica Chimica Acta 378: 311–314. doi:10.1016/j.ica.2011.08.027.
- ↑ Chalkley, Matthew J.; Drover, Marcus W.; Peters, Jonas C. (2020). "Catalytic N2-to-NH3 (or -N2H4) Conversion by Well-Defined Molecular Coordination Complexes". Chemical Reviews 120 (12): 5582–5636. doi:10.1021/acs.chemrev.9b00638. PMID 32352271.
- ↑ 4.0 4.1 Verkade, John G. (1994). "Main group atranes: Chemical and structural features". Coordination Chemistry Reviews 137: 233–295. doi:10.1016/0010-8545(94)03007-D.
- ↑ Milov, Alexey A.; Minyaev, Ruslan M.; Minkin, Vladimir I. (2022). "A DFT insight into the structure and electronic characteristics of group 14 bis-atranes and their analoges". Journal of Organometallic Chemistry 960. doi:10.1016/j.jorganchem.2021.122235.
- ↑ Verkade, John G.; Urgaonkar, Sameer; Verkade, John G.; Urgaonkar, Sameer (2012). "Proazaphosphatrane". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rn00702.pub2. ISBN 978-0471936237.
 |
