Chemistry:Atranorin
From HandWiki
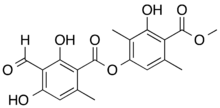
| |
| Names | |
|---|---|
| IUPAC name
(3-hydroxy-4-methoxycarbonyl-2,5-dimethylphenyl) 3-formyl-2,4-dihydroxy-6-methylbenzoate
| |
| Other names | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| Properties | |
| C19H18O8 | |
| Molar mass | 374.345 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Atranorin is a chemical substance produced by some species of lichen. It is a secondary metabolite belonging to a group of compounds known as depsides.[3][4][5] Atranorin has analgesic, anti-inflammatory, antibacterial, antifungal, cytotoxic, antioxidant, antiviral, and immunomodulatory properties.[5][4][6][7] In rare cases, people can have an allergic reaction to atranorin.[8]
References
- ↑ 1.0 1.1 (in en) Atranorin (CAS 479-20-9). https://www.caymanchem.com/product/28106.
- ↑ 479-20-9 | Atranorin | Atranorine; NSC 249980; NSC 685591; NSC 87512; Parmelin; Parmelin Acid; Usnarin; Usnarin Acid | C19G18O8 | TRC. https://www.trc-canada.com/product-detail/?A793810.
- ↑ Melo, Marcelia Garcez Dória; dos Santos, João Paulo Almeida; Serafini, Mairim Russo; Caregnato, Fernanda Freitas; de Bittencourt Pasquali, Matheus Augusto; Rabelo, Thallita Kelly; da Rocha, Ricardo Fagundes; Quintans Jr., Lucindo et al. (March 2011). "Redox properties and cytoprotective actions of atranorin, a lichen secondary metabolite". Toxicology in Vitro 25 (2): 462–468. doi:10.1016/j.tiv.2010.11.014. PMID 21111802.
- ↑ 4.0 4.1 Jaeck, Andreas. Atranorin. https://www.internetchemie.info/stoffdaten/index.php?Datei=Atranorin.
- ↑ 5.0 5.1 Studzinska-Sroka, Elzbieta; Galanty, Agnieszka; Bylka, Wieslawa (7 November 2017). "Atranorin - An Interesting Lichen Secondary Metabolite". Mini-Reviews in Medicinal Chemistry 17 (17): 1633–1645. doi:10.2174/1389557517666170425105727. PMID 28443519.
- ↑ Zhou, Rui; Yang, Yi; Park, So-Yeon; Nguyen, Thanh Thi; Seo, Young-Woo; Lee, Kyung Hwa; Lee, Jae Hyuk; Kim, Kyung Keun et al. (15 August 2017). "The lichen secondary metabolite atranorin suppresses lung cancer cell motility and tumorigenesis" (in en). Scientific Reports 7 (1): 8136. doi:10.1038/s41598-017-08225-1. ISSN 2045-2322. PMID 28811522. Bibcode: 2017NatSR...7.8136Z.
- ↑ Melo, Marcelia Garcez Dória; dos Santos, João Paulo Almeida; Serafini, Mairim Russo; Caregnato, Fernanda Freitas; de Bittencourt Pasquali, Matheus Augusto; Rabelo, Thallita Kelly; da Rocha, Ricardo Fagundes; Quintans, Lucindo et al. (March 2011). "Redox properties and cytoprotective actions of atranorin, a lichen secondary metabolite". Toxicology in Vitro 25 (2): 462–468. doi:10.1016/j.tiv.2010.11.014. PMID 21111802.
- ↑ Allergen Atranorin Allergie Allergologie. https://www.alles-zur-allergologie.de/Allergologie/Artikel/3485/Allergen,Allergie/Atranorin/.
Further reading
- (in de) Zeitschrift Für Naturforschung: A journal of chemical sciences. B. Verlag der Zeitschrift für Naturforschung.
- (in de) Jahresbericht über die Fortschritte der Chemie und verwandter Theile anderer Wissenschaften. Friedrich Vieweg Und Sohn.
- Vos, Carlo; Mckinney, Phillip; Pearson, Colby; Heiny, Erik; Gunawardena, Gamini; Holt, Emily A. (2018). "The optimal extraction and stability of atranorin from lichens, in relation to solvent and pH" (in en). The Lichenologist 50 (4): 499–512. doi:10.1017/S0024282918000075. ISSN 0024-2829.
- Harikrishnan, Adhikesavan; Veena, V.; Lakshmi, B.; Shanmugavalli, R.; Theres, Sonia; Prashantha, C. N.; Shah, Tanya; Oshin, K. et al. (4 March 2021). "Atranorin, an antimicrobial metabolite from lichen Parmotrema rampoddense exhibited in vitro anti-breast cancer activity through interaction with Akt activity". Journal of Biomolecular Structure and Dynamics 39 (4): 1248–1258. doi:10.1080/07391102.2020.1734482. ISSN 0739-1102. PMID 32096436.
 |
