Chemistry:Benzopyran
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H8O | |
| Molar mass | 132.162 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
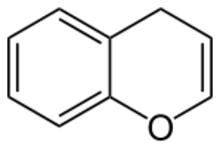
Benzopyran is a polycyclic organic compound that results from the fusion of a benzene ring to a heterocyclic pyran ring.
According to current IUPAC nomenclature, the name chromene used in previous recommendations is retained; however, systematic ‘benzo’ names, for example 2H-1-benzopyran, are preferred IUPAC names for chromene, isochromene, chromane, isochromane, and their chalcogen analogues.[1] There are two isomers of benzopyran that vary by the orientation of the fusion of the two rings compared to the oxygen, resulting in 1-benzopyran (chromene) and 2-benzopyran (isochromene)—the number denotes where the oxygen atom is located by standard naphthalene-like nomenclature.
Some benzopyrans have shown anticancerous activity in vitro.[2]
The radical form of benzopyran is paramagnetic. The unpaired electron is delocalized over the whole benzopyran molecule, rendering it less reactive than one would expect otherwise. A similar example is the cyclopentadienyl radical. Commonly, benzopyran is encountered in the reduced state, in which it is partially saturated with one hydrogen atom, introducing a tetrahedral CH2 group in the pyran ring. Therefore, there are many structural isomers owing to the multiple possible positions of the oxygen atom and the tetrahedral carbon atom:
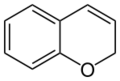 2H-chromene (2H-1-benzopyran) |
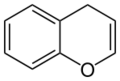 4H-chromene (4H-1-benzopyran) |
| 5H-chromene | 7H-chromene |
| 8aH-chromene |
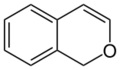 1H-isochromene (1H-2-benzopyran) |
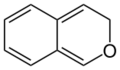 3H-isochromene (3H-2-benzopyran) |
See also
References
- ↑ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. pp. P001–P004. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ Stevenson, Alexander J; Ager, Eleanor I; Proctor, Martina A; Škalamera, Dubravka; Heaton, Andrew; Brown, David; Gabrielli, Brian G (2018). "Mechanism of action of the third generation benzopyrans and evaluation of their broad anti-cancer activity in vitro and in vivo". Scientific Reports 8 (1): 5144. doi:10.1038/s41598-018-22882-w. PMID 29572477. Bibcode: 2018NatSR...8.5144S.
