Chemistry:Benzothiazole
Benzothiazole, or more specifically 1,3-benzothiazole, is an aromatic heterocyclic compound with the chemical formula C7H5NS. It is colorless, slightly viscous liquid. Although the parent compound, benzothiazole is not widely used, many of its derivatives are found in commercial products or in nature. Firefly luciferin can be considered a derivative of benzothiazole.[1] It has a sulfurous odor and meaty flavor.[2]
The three structural isomers of benzothizaole are 1,3-benzothiazole, 1,2-benzothiazole and 2,1-benzothiazole.
Structure and reactivity
Benzothiazoles consist of a 5-membered 1,3-thiazole ring fused to a benzene ring. The nine atoms of the bicycle and the attached substituents are coplanar. The heterocyclic core of the molecule is readily substituted at the methyne (CH) centre in the thiazole ring. Thiazole is electron-withdrawing.
Synthesis and biosynthesis
Benzothiazoles are typically prepared by treatment of 2-mercaptoaniline.[3] For example, acid chlorides are effective:[4]
- C6H4(NH2)SH + RC(O)Cl → C6H4(N)SCR + HCl + H2O
Many other precursors have been used, commonly aldehydes in the presence of oxidants. In some cases, benzothiazoles are prepared directly from anilines, a process that entails ortho functionalization.[5]
Naturally occurring benzothiazoles are proposed to arise by condensation of cysteine with quinones.[1]
Uses
Dyes
The dye thioflavin is a benzothiazole derivative.[6]
Food additives
Benzothiazole occurs naturally in some foods but is also used as a food additive.[1] It has a sulfurous odor and meaty flavor.[7] The European Food Safety Authority assessment had "no safety concern at estimated levels of intake as a flavouring substance".[8]
Rubber additive
Accelerators for the sulfur vulcanization of rubber are based on 2-mercaptobenzothiazoles.[9]
Pharmacology
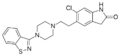
Benzothiazoles have been widely investigated for their bioactivity.[10][11] The benzothiazole moiety is, for example, seen in certain dopamine-acting drugs, e.g. riluzole and pramipexole. Moreover, benzothiazole derivatives act as monoamine oxidase inhibitors or dopamine antagonists:
- 6-oxoethers and derivatives of thiadiazole, thiazolyhydrazine that act selectively on either MAO-A or MAO-B depending on the O-sidechain[12][13][14]
- 1-alkylpiperidines that act on dopamine D4 receptor[15][16]
See also
- thiazoles, which lack the fused benzene ring.
- Benzoxazoles, which substitute an oxygen for the sulfur atom.
- 2-Aminobenzothiazoles, well-studied derivatives of benzothiazole
Safety and environmental considerations
Benzothiazoles are widely used in the vulcanization of rubber, so their possible role in the environment has attracted attention. Evidence suggests that they biodegrade readily.[17]
References
- ↑ 1.0 1.1 1.2 Le Bozec, Lucille; Moody, Christopher J. (2009). "Naturally Occurring Nitrogen–Sulfur Compounds. The Benzothiazole Alkaloids". Australian Journal of Chemistry 62 (7): 639. doi:10.1071/CH09126.
- ↑ "Benzothiazole". http://www.thegoodscentscompany.com/data/rw1007521.html.
- ↑ Gill, Rupinder K.; Rawal, Ravindra K.; Bariwal, Jitender (2015). "Recent Advances in the Chemistry and Biology of Benzothiazoles". Archiv der Pharmazie 348 (3): 155–178. doi:10.1002/ardp.201400340. PMID 25682746.
- ↑ T. E. Gilchrist "Heterocyclic Chemistry" 3rd Edition, Longman, 1992.
- ↑ Würfel, Hendryk; Jakobi, Dörthe (2018). "Syntheses of Substituted 2-Cyano-benzothiazoles". Organic Syntheses 95: 177–191. doi:10.15227/orgsyn.095.0177.
- ↑ Gill, Rupinder K.; Rawal, Ravindra K.; Bariwal, Jitender (2015). "Recent Advances in the Chemistry and Biology of Benzothiazoles". Archiv der Pharmazie 348 (3): 155–178. doi:10.1002/ardp.201400340. PMID 25682746.
- ↑ "Benzothiazole". http://www.thegoodscentscompany.com/data/rw1007521.html.
- ↑ "Flavouring Group Evaluation 76, (FGE.76) - Consideration of sulphur-containing heterocyclic compounds evaluated by JECFA (59th meeting) structurally related to thiazoles, thiophene, thiazoline and thienyl derivatives from chemical group 29, miscellaneous". EFSA Journal 6 (11): 875. 2008. doi:10.2903/j.efsa.2008.875.
- ↑ Engels, Hans-Wilhelm; Weidenhaupt, Herrmann-Josef; Pieroth, Manfred; Hofmann, Werner; Menting, Karl-Hans; Mergenhagen, Thomas; Schmoll, Ralf; Uhrlandt, Stefan (2004), "Rubber, 4. Chemicals and Additives", Ullmann's Encyclopedia of Industrial Chemistry, doi:10.1002/14356007.a23_365.pub2, ISBN 3-527-30673-0
- ↑ Rouf, Abdul; Tanyeli, Cihangir (2015). "Bioactive thiazole and benzothiazole derivatives". European Journal of Medicinal Chemistry 97: 911–927. doi:10.1016/j.ejmech.2014.10.058. PMID 25455640.
- ↑ Keri, Rangappa S.; Patil, Mahadeo R.; Patil, Siddappa A.; Budagumpi, Srinivasa (2015). "A comprehensive review in current developments of benzothiazole-based molecules in medicinal chemistry". European Journal of Medicinal Chemistry 89: 207–251. doi:10.1016/j.ejmech.2014.10.059. PMID 25462241.
- ↑ le Roux, Anandie; Petzer, Anél; Cloete, Stephanus J.; Petzer, Jacobus P. (2025-03-01). "An investigation of the monoamine oxidase inhibition properties of benzothiazole derivatives". Results in Chemistry 14. doi:10.1016/j.rechem.2025.102142. ISSN 2211-7156.
- ↑ Acar Çevik, Ulviye; Osmaniye, Derya; Sağlik, Begüm N.; Levent, Serkan; K. Çavuşoğlu, Betül; Karaduman, Abdullah B.; D. Özkay, Ümide; Özkay, Yusuf et al. (2020). "Synthesis of new benzothiazole derivatives bearing thiadiazole as monoamine oxidase inhibitors" (in en). Journal of Heterocyclic Chemistry 57 (5): 2225–2233. doi:10.1002/jhet.3942. ISSN 1943-5193. https://onlinelibrary.wiley.com/doi/10.1002/jhet.3942.
- ↑ Turan, Gülhan; Osmaniye, Derya; Sağlik, Begüm Nurpelin; Çevik, Ulviye Acar; Levent, Serkan; Çavuşoğlu, Betül Kaya; Özkay, Ümide Demir; Özkay, Yusuf et al. (2020-06-02). "Synthesis and monoamine oxidase A/B inhibitory evaluation of new benzothiazole-thiazolylhydrazine derivatives". Phosphorus, Sulfur, and Silicon and the Related Elements 195 (6): 491–497. doi:10.1080/10426507.2020.1722667. ISSN 1042-6507. https://www.tandfonline.com/doi/full/10.1080/10426507.2020.1722667.
- ↑ Boateng, Comfort A.; Nilson, Ashley N.; Placide, Rebekah; Pham, Mimi L.; Jakobs, Franziska M.; Boldizsar, Noelia; McIntosh, Scot; Stallings, Leia S. et al. (2023-09-14). "Pharmacology and Therapeutic Potential of Benzothiazole Analogues for Cocaine Use Disorder". Journal of Medicinal Chemistry 66 (17): 12141–12162. doi:10.1021/acs.jmedchem.3c00734. ISSN 0022-2623. PMID 37646374.
- ↑ Sampson, Dinithia; Zhu, Xue Y.; Eyunni, Suresh V. K.; Etukala, Jagan R.; Ofori, Edward; Bricker, Barbara; Lamango, Nazarius S.; Setola, Vincent et al. (2014-06-15). "Identification of a new selective dopamine D4 receptor ligand". Bioorganic & Medicinal Chemistry 22 (12): 3105–3114. doi:10.1016/j.bmc.2014.04.026. ISSN 0968-0896. PMID 24800940.
- ↑ Clarke, Bradley O.; Smith, Stephen R. (2011). "Review of 'emerging' organic contaminants in biosolids and assessment of international research priorities for the agricultural use of biosolids". Environment International 37 (1): 226–247. doi:10.1016/j.envint.2010.06.004. PMID 20797791. Bibcode: 2011EnInt..37..226C.
External links
Template:Simple aromatic rings
 |
