Chemistry:Blaise ketone synthesis
| Blaise ketone synthesis | |
|---|---|
| Named after | Edmond Blaise |
| Reaction type | Coupling reaction |
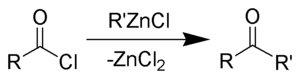
The Blaise ketone synthesis (named after Edmond Blaise) is the chemical reaction of acid chlorides with organozinc compounds to give ketones.[1][2]
The reaction was claimed to bring excellent yields by Blaise, however, investigators failed to obtain better than moderate yields (50%).[3][4] Thus, the reaction is particularly ineffective in forming ketones from acyl chlorides. The reaction also works with organocuprates.[5][6] Reviews have been written.[7][8]
Reaction mechanism
The mechanism is sampled from the proposed mechanism for organocadmium compounds, given that the mechanisms are identical to one another the proposed mechanism for the reaction is the same as the one for organocadmium compounds[9][10].
After the oxygen forms a bond with the organozinc compound, R’ shifts to the carbonyl carbon, having chlorine act as a leaving group and removing the negative charge from zinc. The chlorine that left returns to form a bond with zinc, pushing the electrons back on to oxygen and thus forming the ketone.[11]
Variations
Blaise-Maire reaction
The Blaise-Maire reaction is the Blaise ketone synthesis using β-hydroxy acid chlorides to give β-hydroxyketones, which are converted into α,β-unsaturated ketones using sulfuric acid.[12]
Ketone formation from organocadmium compounds
This ketone formation is an identical reaction to the Blaise ketone synthesis. Only instead of organozinc compounds, organocadmium compounds are used and produce higher yields.
See also
References
- ^ Blaise, E. E.; Koehler, A. (1910). "Synthèse au moyen des dérivés organo-métalliques mixtes du zinc (II).". Bull. Soc. Chim. Fr. 7: 215–227.
- ^ Blaise, E. E. (1911). "Sur les dérivés organo-métalliques mixtes du zinc et leur emploi dans la synthèse organique". Bull. Soc. Chim. Fr. 9: 1.
- ^ Posner, G. H.; Whitten, C. E. (1976). "Secondary and Tertiary Alkyl Ketones from Carboxylic Acid Chlorides and Lithium Phenylthio(Alkyl)Cuprate Reagents: tert-Butyl Phenyl Ketone". Organic Syntheses 55: 122. http://www.orgsyn.org/demo.aspx?prep=cv6p0248.; Collective Volume, 6, pp. 248
- ^ Fujisawa, T.; Sato, T. (1988). "Ketones from Carboxylic Acids and Grignard Reagents: Methyl 6-Oxodecanoate". Organic Syntheses 66: 116. http://www.orgsyn.org/demo.aspx?prep=cv8p0441.; Collective Volume, 8, pp. 441
- ^ Cason, J. (1947). "The Use of Organocadmium Reagents for the Preparation of Ketones". Chem. Rev. 40 (1): 17. doi:10.1021/cr60125a002. PMID 20287882.
- ^ Shirley, D. A. (1954). "The Synthesis of Ketones from Acid Halides and Organometallic Compounds of Magnesium, Zinc, and Cadmium". Organic Reactions 8: 29. doi:10.1002/0471264180.or008.02. ISBN 0471264180.
- ^ Blaise, E. E.; Maire, M. (1907). "Synthèse au moyen des dérivés organo-métalliques mixtes du zinc. Cétones non saturées αβ-acycliques". Compt. Rend. 145: 73-75. https://gallica.bnf.fr/ark:/12148/bpt6k3099v/f73.image.
- ^ Iqbal, M.; Baloch, I. B.; Baloch, K. (2012). "An efficient method for the preparation of benzyl γ-ketohexanoates.". Chemical Papers-Slovak Academy of Sciences: 10. doi:10.2478/s11696-012-0282-8.
- ^ Vogel, A. I. (1989). Vogel's Textbook of Practical Organic Chemistry (5 ed.). Prentice Hall. pp. 33-34. ISBN 0-582-46236-3. https://faculty.ksu.edu.sa/sites/default/files/vogel_-_practical_organic_chemistry_5th_edition.pdf.
 |