Chemistry:Bromopentaamminecobalt(III) bromide
From HandWiki
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| |
| |
| Properties | |
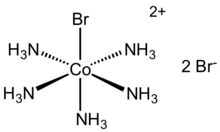
| [Co(NH3)5Br]Br2 | |
| Molar mass | 383.798 g/mol |
| Appearance | Purple |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Bromopentaamminecobalt(III) bromide is the dibromide salt of the cobalt coordination compound with the formula [Co(NH3)5Br]2+. It is a purple, water-soluble solid. The analogous chloropentaamminecobalt(III) chloride is also well known.
Synthesis and reactions
The title compound is prepared by oxidation of a solution of cobalt(II) salts in aqueous ammonia.[1]
- 2 CoBr2 + 8 NH3 + 2 NH4Br + H2O2 → 2 [Co(NH3)5Br]Br2 + 2 H2O
It was first reported in the 1870s, before the structure or even formulae were understood for such complexes. This early work showed that only two thirds of the bromide groups were exchangeable with other anions such as nitrate and dithionate.[2]
The complex undergoes aquation, meaning that bromide is displaced by water:
- [Co(NH3)5Br]Br2 + H2O → [Co(NH3)5(H2O)]Br3
This process is catalyzed by platinum.[3]
References
- ↑ Diehl, Harvey; Clark, Helen; Willard, H. H. (1939). "Bromopentamminocobalti Bromide". Inorganic Syntheses. Inorganic Syntheses. 1. pp. 186–188. doi:10.1002/9780470132326.ch66. ISBN 9780470132326.
- ↑ Jörgensen, S. M. (1879). "Beiträge zur Chemie der Kobaltammoniakverbindungen. II. Ueber die Bromopurpureokobaltsalze". Journal für Praktische Chemie 19: 49–69. doi:10.1002/prac.18790190110. https://zenodo.org/record/1427878.
- ↑ Archer, M. D.; Spiro, M. (1970). "Heterogeneous Catalysis in Solution. Part VIII. Catalysis of the Aquation of the Bromopentaamminecobalt(III) Ion by Metallic Platinum". Journal of the Chemical Society (1): 78–81. doi:10.1039/j19700000078.
Further reading
- Loehlin, James H. "The Study of a Cobalt Complex-A Laboratory Project." Journal of Chemical Education. doi:10.1021/ed059p1048
- Williams, Gregory M; Olmsted, John, III; Preksa, Andrew P., III (1989). "Coordination complexes of cobalt: inorganic synthesis in the general chemistry laboratory". Journal of Chemical Education 66 (12): 1043–5. doi:10.1021/ed066p1043. Bibcode: 1989JChEd..66.1043W.
 |

