Chemistry:Cll1
Toxin Cll1 is a toxin from the venom of the Mexican scorpion Centruroides limpidus limpidus, which changes the activation threshold of sodium channels by binding to neurotoxin binding site 4, resulting in increased excitability.
Etymology and source
The toxin Cll1 is named after its producing species, Centruroides limpidus limpidus. Along with Cll1, multiple toxins are excreted in its venom.[2]
Chemistry
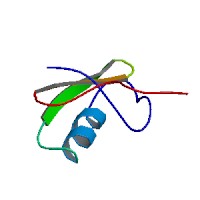
Cll1 is a long chain neuropeptide belonging to the scorpion toxin superfamily. Cll1 is classified as a member of the beta-toxin subfamily.[3] The global secondary structure of Cll1 is similar to that of other scorpion beta-toxins, including the alpha-helix, triple stranded antiparallel beta-sheet, and the four disulfide bridges.[4][5] The higher affinity for crustacean rather than mammalian sodium channels has been attributed to the presence of Trp18, a hydrophobic amino acid at the surface of Cll1.[4]
Target
Cll1 targets, like the classical scorpion beta-toxin, the voltage-gated sodium channels (Nav). Beta-toxins bind to the extracellular end of the voltage sensor S4 at the loop between the 3rd and 4th segment of the second domain.[6] By binding it alters the voltage dependent opening of the channel.[7]
Mode of action
Cll1 influences three intrinsic properties of the targeted sodium channel:
Voltage dependent activation
Cll1 binds to transmembrane segment S4 of the voltage gated sodium channels. Its binding shifts the activation threshold of the sodium channel towards more negative membrane potentials.[7] Seven different isoforms of the voltage gated sodium channels (Nav1.1-Nav1.7) have been studied in the presence of Cll1. In almost all of these seven isoforms, Cll1 affects voltage dependent activation. It has only a minor effect on the Nav1.1-1.4 and Nav1.7 channels, but a much larger effect on isoform Nav1.6.[7]
Peak current
Cll1 causes a reduction of the peak current when bound to the voltage activated sodium channels. This effect was present in all but one of the seven tested isoforms (Nav1.1-Nav1.6). The only isoform that showed no reduction in peak current was Nav1.7.[7]
Resurgent current
Cll1 can induce resurgent currents. This effect has also been demonstrated for other beta-scorpion toxins. The resurgent current is strongest in Nav1.6, but it is also present to a much lesser extent in other isoforms of the voltage activated sodium channels.[7]
Toxicity and treatment
The LD50 of the Cll1 toxin in mice is 85 μg/kg.[8] A possible treatment for an intoxication by Cll1 toxin is the use of single chain variable fragments (scFv).[8] Other possible treatments find their origin in traditional Mexican medicine. Several herbs used in traditional Mexican medicine have been proven to be effective in treating an intoxication from the whole venom from C. limpidus limpidus in mice, including Bouvardia ternifolia.[9]
References
- ↑ "TR SCXR_CENLL". https://modbase.compbio.ucsf.edu/modbase-cgi/model_details.cgi?queryfile=1444655752_5506&searchmode=default&displaymode=moddetail&referer=yes&snpflag=&.
- ↑ Dehesa-Dávila, M; Possani, LD (September 1994). "Scorpionism and serotherapy in Mexico.". Toxicon 32 (9): 1015–8. doi:10.1016/0041-0101(94)90383-2. PMID 7801335. Bibcode: 1994Txcn...32.1015D.
- ↑ "UniProtKB - P45667 (SCXR_CENLL)". https://www.uniprot.org/uniprot/P45667#structure. Retrieved 12 October 2015.
- ↑ 4.0 4.1 Lebreton, F; Delepierre, M; Ramírez, AN; Balderas, C; Possani, LD (20 September 1994). "Primary and NMR three-dimensional structure determination of a novel crustacean toxin from the venom of the scorpion Centruroides limpidus limpidus Karsch.". Biochemistry 33 (37): 11135–49. doi:10.1021/bi00203a010. PMID 7727365.
- ↑ Zhijian, C; Yingliang, W; Jiqun, S; Wanhong, L; Fan, X; Xin, M; Hui, L; Dahe, J et al. (2003). "Evidence for the existence of a common ancestor of scorpion toxins affecting ion channels.". Journal of Biochemical and Molecular Toxicology 17 (4): 235–8. doi:10.1002/jbt.10083. PMID 12898647.
- ↑ Cestèle, S; Qu, Y; Rogers, JC; Rochat, H; Scheuer, T; Catterall, WA (October 1998). "Voltage sensor-trapping: enhanced activation of sodium channels by beta-scorpion toxin bound to the S3-S4 loop in domain II.". Neuron 21 (4): 919–31. doi:10.1016/S0896-6273(00)80606-6. PMID 9808476.
- ↑ 7.0 7.1 7.2 7.3 7.4 Schiavon, E; Pedraza-Escalona, M; Gurrola, GB; Olamendi-Portugal, T; Corzo, G; Wanke, E; Possani, LD (February 2012). "Negative-shift activation, current reduction and resurgent currents induced by β-toxins from Centruroides scorpions in sodium channels.". Toxicon 59 (2): 283–93. doi:10.1016/j.toxicon.2011.12.003. PMID 22200496. Bibcode: 2012Txcn...59..283S.
- ↑ 8.0 8.1 Riaño-Umbarila, L; Olamendi-Portugal, T; Morelos-Juárez, C; Gurrola, GB; Possani, LD; Becerril, B (15 December 2013). "A novel human recombinant antibody fragment capable of neutralizing Mexican scorpion toxins.". Toxicon 76: 370–6. doi:10.1016/j.toxicon.2013.09.016. PMID 24067940. Bibcode: 2013Txcn...76..370R.
- ↑ Jiménez-Ferrer, JE; Pérez-Terán, YY; Román-Ramos, R; Tortoriello, J (January 2005). "Antitoxin activity of plants used in Mexican traditional medicine against scorpion poisoning.". Phytomedicine 12 (1–2): 116–22. doi:10.1016/j.phymed.2003.10.001. PMID 15693718.
 |
