Chemistry:D-DOPA
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
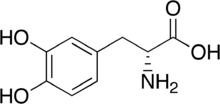
3,4-Dihydroxy-D-phenylalanine
| |
| Systematic IUPAC name
(2R)-2-Amino-3-(3,4-dihydroxyphenyl)propanoic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H11NO4 | |
| Molar mass | 197.19 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
d-DOPA (D-3,4-dihydroxyphenylalanine; dextrodopa) is similar to L-DOPA (levodopa), but with opposite chirality.[1] Levo- and dextro- rotation refer to a molecule's ability to rotate planes of polarized light in one or the other direction. Whereas L-DOPA is moderately effective in the treatment of Parkinson's disease (PD) and dopamine-responsive dystonia (DRD) by stimulating the production of dopamine in the brain, D-DOPA is biologically inactive.
Avoiding unwanted production of D-DOPA is a goal for commercial synthesis of L-DOPA-based drugs to treat Parkinson's disease.[1]
See also
- l-DOPA (Levodopa; Sinemet, Parcopa, Atamet, Stalevo, Madopar, Prolopa, etc.)
- l-DOPS (Droxidopa)
- Methyldopa (Aldomet, Apo-Methyldopa, Dopamet, Novomedopa, etc.)
- Dopamine (Intropan, Inovan, Revivan, Rivimine, Dopastat, Dynatra, etc.)
- Norepinephrine (Noradrenaline; Levophed, etc.)
- Epinephrine (Adrenaline; Adrenalin, EpiPen, Twinject, etc.)
References
- ↑ 1.0 1.1 Kolb, Vera M. (2017-04-21) (in en). Green Organic Chemistry and its Interdisciplinary Applications. CRC Press. pp. 130. ISBN 978-1-4987-0208-9. https://www.google.com/books/edition/Green_Organic_Chemistry_and_its_Interdis/hRdjDAAAQBAJ?hl=en&gbpv=1&dq=d-DOPA+chiral&pg=PA130&printsec=frontcover.
 |
