Chemistry:Dibromoiodoethylene
From HandWiki
| |||
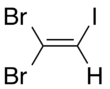 1,1- isomer
| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol)
|
| ||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C2HBr2I | |||
| Molar mass | 311.742 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
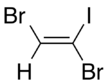
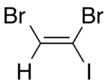
Dibromoiodoethylene is a dense organic liquid consisting of an ethylene core with two bromine atoms and one iodine atom as substituents. It is formed by the reaction of dibromoacetylene with hydrogen iodide.[1]
There are three isomers of dibromoiodoethylene, varying in the position of the two bromine atoms compared to each other.
References
- ↑ Sartori, Mario (1939). The War Gases Chemistry and Analysis. New York: D. Van Nostrand Co., Inc.. p. 51. https://archive.org/stream/The_War_Gases_Chemistry_and_Analysis_Sartori/The_War_Gases_Chemistry_and_Analysis_Sartori_djvu.txt.
 |