Chemistry:Larock indole synthesis
| Larock indole synthesis | |
|---|---|
| Named after | Richard C. Larock |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000396 |
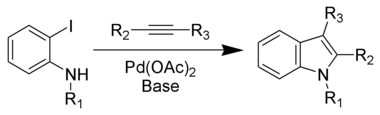
The Larock indole synthesis is a heteroannulation reaction that uses palladium as a catalyst to synthesize indoles from an ortho-iodoaniline and a disubstituted alkyne.[1] It is also known as Larock heteroannulation. The reaction is extremely versatile and can be used to produce varying types of indoles. Larock indole synthesis was first proposed by Richard C. Larock in 1991 at Iowa State University.[2]
Overall reaction
The reaction usually occurs with an o-iodianiline or its derivatives, 2–5 equivalents of an alkyne, palladium(II) (PdII), an excess of sodium or potassium carbonate base, PPh3, and 1 equivalent of LiCl or n-Bu4NCl. N-methyl, N-acetyl, and N-tosyl derivatives of ortho-iodoanilines have been shown to be the most successful anilines that can be used to produce good to excellent yields.[3]
Reagents and optimal conditions
Chlorides
Either LiCl or n-Bu4N are used depending on the reaction conditions, but LiCl appears to be the more effective base in Larock indole annulation.[3] The stoichiometry of LiCl is also considerably important, as more than 1 equivalent of LiCl will slow the rate of reaction and lower the overall yield.[1]
Bases
Bases other than sodium or potassium carbonate have been used to produce a good overall yield of the annulation reaction.[3] For example, KOAc can be used with 1 equivalent of LiCl. However, the reaction using KOAc must be used at 120 °C to reach completion of the reaction at a reasonable time. In contrast K2CO3 can be used at 100 °C.
Alkynes
The Larock indole synthesis is a flexible reaction partly due to the variety of substituted alkynes that can be used in the annulation reaction. In particular, alkynes with substituents including alkyls, aryls, alkenyls, hydroxyls, and silyls have been successfully used.[3] However, bulkier tertiary alkyl or trimethylsilyl groups have been shown to provide a higher yield.[1] The annulation reaction will also proceed more efficiently when 2–5 equivalents of an alkyne is used. Less than two equivalents appear to create suboptimal conditions for the reaction.
PPh3 as a catalyst
5% mol of PPh3 was initially used in the reaction as a catalyst.[1] However, later experiments have shown that PPh3 does not significantly improve the overall yield and is not necessary.[3]
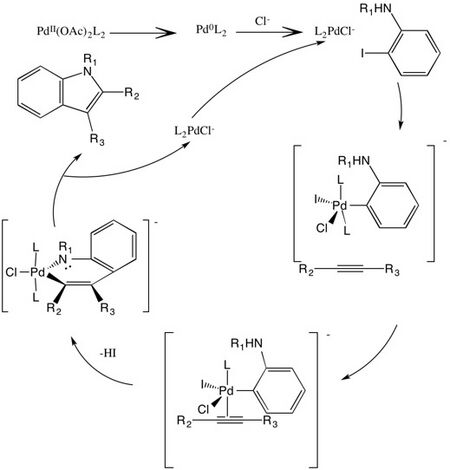
Reaction mechanism
The Larock indole synthesis proceeds via the following intermediate steps:[3]
- Pd(OAc)2 is reduced to Pd(0).
- A coordination of the chloride occurs to form a chloride-ligated zerovalent palladium.
- The o-iodoaniline undergoes oxidative addition to Pd(II).
- The alkyne coordinates to the Pd(II) by ligand exchange.
- A migratory insertion causes the alkyne to undergo regioselective syn-insertion into arylpalladium bond. Regioselectivity is determined during this step.
- The nitrogen displaces the halide in the resulting vinylic palladium intermediate to form the six-membered palladium-containing heteroatom.
- The Pd(II) center undergoes a reductive elimination to form the indole and regenerate Pd(0) which can then be recycled into the catalytic indole process.
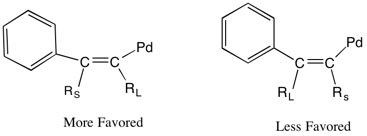
The carbopalladation step is regioselective when unsymmetrical alkynes are used.[1][3] Although it was previously believed that the alkyne is inserted with the less sterically-hindering R-group adjacent to the arylpalladium, Larock et al. observed that the larger more sterically-hindering R-group is inserted next to the arylpalladium.[1] They suggest that the driving force of the alkyne insertion may be the steric hindrance present in the developing carbon-carbon bond and the orientation of the alkyne prior to syn-insertion of the alkyne into the aryl palladium bond.[3] Alkyne insertion occurs so that the large substituent on the alkyne avoids steric strain from the short developing carbon-carbon bond by interacting with the longer carbon-palladium bond.
Modifications and variations
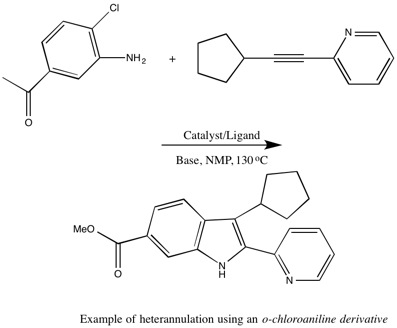
o-bromoanilines or o-chloroanilines do not undergo Larock indole synthesis. However, researchers from Boehringer-Ingelheim were able to successfully use both o-bromoanilines and o-chloroanilines to form indoles by using N-methyl-2-pyrrolidone (NMP) as the solvent with 1,1'bis(di-tert-butylphosphino)ferrocene as the palladium ligand.[4] O-bromoanilines and o-chloroanilines are more readily available and cost-effective over using o-iodianiline in Larock indole synthesis.[2]
Monguchi et al. also derived 2- and 2,3-substituted indoles without using LiCl.[5] The optimized Indole reaction uses 10% Pd/C (3.0 mol%) with 1.1 equivalent of NaOAc, and NMP at 110–130 °C. Monguchi et al. state that their optimized condition of the Larock indole synthesis without LiCl is a more mild, environmentally benign, and efficient strategy for producing indoles.
Applications
Indoles are one of the most prevalent heterocyclic structures found in biological processes, so the production of indole derivatives are important in a diversity of fields.
Nishikawa et al. derived iso-tryptophan by using Larock indole synthesis with pre-synthesized α-C-glucosylpropargyl glycine and o-iodo-tosylanilide.[6] This reaction produced the product which had the reverse regioselectivity of normal Larock indole synthesis. The larger substituent was placed adjacent to the forming carbon-carbon bond, rather than the carbon-palladium bond. The explanation for the reverse regioselectivity which produced the iso-tryptophan is unknown.
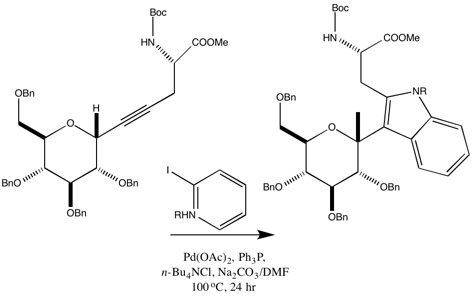
Optically active tryptophan which adheres to the regioselectivity of the Larock indole synthesis can also be synthesized using o-iodoaniline with propargyl substituted bislactim ethyl ether. Propargyl substituted bislactim ethyl ether is generated by using Schöllkopf chiral auxiliary bis lactam ether with n-BuLi, THF, and 3-halo-1-9trimethylsily1)-1-propyne and extracting the trans-isomer of the propargyl-substituted bislactim.[7]

Other relevant applications include the synthesis of 5-HT1D receptor agonist MK-0462, an anti-migraine drug.[8]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Larock, R. C.; Yum, E. K. (1991). "Synthesis of indoles via palladium-catalyzed heteroannulation of internal alkynes". Journal of the American Chemical Society 113 (17): 6689. doi:10.1021/ja00017a059.
- ↑ 2.0 2.1 Li, J.J. (2011) "Larock Indole Synthesis" in Name Reactions in Heterocyclic Chemistry II, John Wiley & Sons, ISBN:978-0-470-08508-0, pp. 143–166.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Larock, R. C.; Yum, E. K.; Refvik, M. D. (1998). "Synthesis of 2,3-Disubstituted Indoles via Palladium-Catalyzed Annulation of Internal Alkynes". The Journal of Organic Chemistry 63 (22): 7652. doi:10.1021/jo9803277.
- ↑ 4.0 4.1 Li, G., Liu, J., Lu, B., Roschangar, F., Senanayake, C.H., Shen, M. (2005) "By reacting with substituted acetylenes in the presence of a phosphine ligand and a base (K2CO3); used to make drugs" U.S. Patent 20,050,209,465
- ↑ Monguchi, Y.; Mori, S.; Aoyagi, S.; Tsutsui, A.; Maegawa, T.; Sajiki, H. (2010). "Palladium on carbon-catalyzed synthesis of 2- and 2,3-substituted indoles under heterogeneous conditions". Organic & Biomolecular Chemistry 8 (14): 3338–42. doi:10.1039/c004939e. PMID 20526486.
- ↑ 6.0 6.1 Nishikawa, T.; Wada, K.; Isobe, M. (2002). "Synthesis of Novel .ALPHA.-C-Glycosylamino Acids and Reverse Regioselectivity in Larock's Heteroannulation for the Synthesis of the Indole Nucleus". Bioscience, Biotechnology, and Biochemistry 66 (10): 2273–8. doi:10.1271/bbb.66.2273. PMID 12450149.
- ↑ 7.0 7.1 Ma, C.; Liu, X.; Yu, S.; Zhao, S.; Cook, J. M. (1999). "Concise synthesis of optically active ring-A substituted tryptophans". Tetrahedron Letters 40 (4): 657. doi:10.1016/S0040-4039(98)02497-6.
- ↑ Chen, C.-Y, Lieberman, D.R., Larsen, R.D., Reamer, R.A., Verhoeven, T.R., Reider, P.J., Cottrell, I.F., Houghton, P.G. (1994). "Synthesis of the 5-HT1D agonist MK-0462 via a Pd-catalyzed coupling reaction". Tetrahedron Lett. 35: 6981–6984. doi:10.1016/0040-4039(94)88204-5.
 |