Chemistry:LmKTT-1a
LmKTT-1a (SdPI-2, δ-KTx2.1) is a bifunctional Kunitz-type toxin belonging to the ẟ-KTx subfamily, which can be found in the venom of Lychas mucronatus (the Chinese swimming scorpion). As a bifunctional toxin, it both inhibits trypsin activity and blocks Kv1 channels with a weak selectivity towards Kv1.3 channels.
Source and etymology
The LmKTT-1a toxin was first identified in Lychas mucronatus, a scorpion belonging to the Buthidae family, which is widely spread around Southeast Asia and southern China.[2]
The LmKTT-1a nomenclature describes the toxin species of origin (Lm: L.mucronatus) and its structural scaffold (KTT: Kunitz-type toxin).
LmKTT-1a was first identified and named as a scorpion-derived protease inhibitor (SdPI-2). The mature protein of this toxin has 96.6% sequence homology with a toxin named LmKTT-1b (SdPI).[3] The mature protein is established by removing the signal sequence prior to it (see Amino Acid Sequence table).[4] The mature proteins of the two toxins differ only in their second and twenty sixth amino acids: SdPI possesses an asparagine (Asn) and a glycine (Gly), whereas SdPI-2 has a lysine (Lys) and a serine (Ser) in these locations, respectively.[3][5]
Chemistry
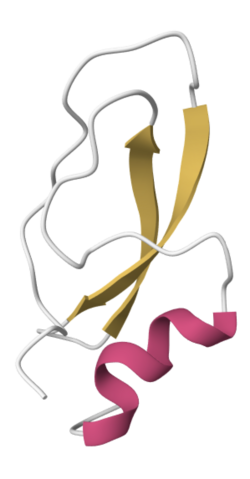
Structure
The mature protein of LmKTT-1a (SdPI-2) is composed of 59 amino acids with a molecular mass of 8658.6 Da.[5][6] These residues assemble into a unique Kunitz-type structural fold, which is typical for the δ-KTx subfamily.[5] This characteristic fold consists of a short α-helix connected to two antiparallel β-sheets with six cysteines in the sequence to form three disulfide bridges (see figure 1).[5][7] This ẟ-KTx subfamily's typical Kunitz-type fold represents a third possible structure of scorpion toxins specific to potassium (K+) channels (KTx).[5]
|
MKSFLLIALVLFFLFVSYASAKKKCQLPSDVGKGKASFTRYYYNEESGKCETFIYGGVGGNSNNFLTKEDCCRECAQGSC Signal Sequence Mature Protein (1-59) |
Cysteine framework
LmKTT-1a adopts a distinctive cysteine framework.[5] While Kunitz-type toxins normally have a CysII-CysIV disulfide bridge, LmKTT-1a lacks this bridge and instead possesses two cysteine residues near the C-terminus (Cys51 and Cys59), which form a new disulfide bridge.[5][6] This disulfide bridge has little to no effect on the toxin's fold and ability to block potassium channels.[5] Yet, after eliminating this Cys51-C59 disulfide bridge, trypsin was inhibited with five-fold lower Ki than wildtype LMKTT-1a.[5]
Targets
LmKTT-1a targets voltage-gated potassium channels and blocks them. The toxin has a weak selectivity for Kv1.3 channels with an IC50-value of 1.58±0.73 µM, with less effect on Kv1.1 and Kv1.2 channels.[5]
Moreover, LmKTT-1a also selectively inhibits trypsin with a Ki value of 0.14-0.16 µM at a 1:1 stoichiometric ratio, while not affecting chymotrypsin and elastase activity.[3][5]
Mode of Action
The related toxin LmKTT-1b (SdPI) inhibits trypsin activity through forming a complex with trypsin, where LmKTT-1b's active site (K12 to A15) interacts with the S1 pocket of trypsin.[3] In this pocket, K14 forms hydrogen bonds with two residues of trypsin (D176 and S192). This K14 is crucial for LmKTT-1b's inhibitory activity, as this activity is fully abolished when K14 is mutated to a hydrophobic residue.[3]
References
- ↑ "3D View: 2M01". https://www.rcsb.org/3d-view/2M01.
- ↑ Ruiming, Zhao; Yibao, Ma; Yawen, He; Zhiyong, Di; Yingliang, Wu; Zhijian, Cao; Wenxin, Li (2010-07-28). "Comparative venom gland transcriptome analysis of the scorpion Lychas mucronatus reveals intraspecific toxic gene diversity and new venomous components". BMC Genomics 11 (1): 452. doi:10.1186/1471-2164-11-452. ISSN 1471-2164. PMID 20663230.
- ↑ 3.0 3.1 3.2 3.3 3.4 Zhao, Ruiming; Dai, Hui; Qiu, Su; Li, Tian; He, Yawen; Ma, Yibao; Chen, Zongyun; Wu, Yingliang et al. (2011-11-08). "SdPI, The First Functionally Characterized Kunitz-Type Trypsin Inhibitor from Scorpion Venom". PLOS ONE 6 (11): e27548. doi:10.1371/journal.pone.0027548. ISSN 1932-6203. PMID 22087336. Bibcode: 2011PLoSO...627548Z.
- ↑ 4.0 4.1 "UniProt". https://www.uniprot.org/uniprotkb/P0DJ46/entry.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 Chen, Zongyun; Luo, Fan; Feng, Jing; Yang, Weishan; Zeng, Danyun; Zhao, Ruiming; Cao, Zhijian; Liu, Maili et al. (2013-04-03). "Genomic and Structural Characterization of Kunitz-Type Peptide LmKTT-1a Highlights Diversity and Evolution of Scorpion Potassium Channel Toxins". PLOS ONE 8 (4): e60201. doi:10.1371/journal.pone.0060201. ISSN 1932-6203. PMID 23573241. Bibcode: 2013PLoSO...860201C.
- ↑ 6.0 6.1 Chen, Zong-Yun; Hu, You-Tian; Yang, Wei-Shan; He, Ya-Wen; Feng, Jing; Wang, Bin; Zhao, Rui-Ming; Ding, Jiu-Ping et al. (April 2012). "Hg1, Novel Peptide Inhibitor Specific for Kv1.3 Channels from First Scorpion Kunitz-type Potassium Channel Toxin Family". Journal of Biological Chemistry 287 (17): 13813–13821. doi:10.1074/jbc.m112.343996. ISSN 0021-9258. PMID 22354971.
- ↑ Zweckstetter, Markus; Czisch, Michael; Mayer, Ulrike; Chu, Mon-Li; Zinth, Wolfgang; Timpl, Rupert; Holak, Tad A (February 1996). "Structure and multiple conformations of the Kunitz-type domain from human type VI collagen α3(VI) chain in solution". Structure 4 (2): 195–209. doi:10.1016/S0969-2126(96)00022-6.
 |

