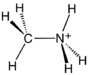
Chemistry:Methylammonium iodide
From HandWiki
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Methylazanium iodide
| |||
| Systematic IUPAC name
Methanaminium iodide | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| CH3NH3I | |||
| Molar mass | 158.96951 g/mol | ||
| Appearance | White powder [1] | ||
| Hazards | |||
| GHS pictograms | 
| ||
| GHS Signal word | Warning | ||
| H302, H315, H319, H335 | |||
| P261, P264, P270, P271, P280, P301+312, P302+352, P304+340, P305+351+338, P312, P321, P330, P332+313, P337+313, P362, P403+233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
Methylammonium iodide in an organic halide with a formula of CH3NH3I. It is an ammonium salt composed of methylamine and hydrogen iodide. The primary application for methylammonium iodide, sometimes in combination with other methylammonium halides, is as a component of perovskite (structure) crystalline solar cells.[2]
References
- ↑ "Methylammonium Iodide". https://www.greatcellsolarmaterials.com/methylammonium-iodide.html.
- ↑ Li, Hangqian. (2016). "A modified sequential deposition method for fabrication of perovskite solar cells". Solar Energy 126: 243–251. doi:10.1016/j.solener.2015.12.045. Bibcode: 2016SoEn..126..243L.
 |