Chemistry:Michellamine
| |
| Names | |
|---|---|
| Preferred IUPAC name
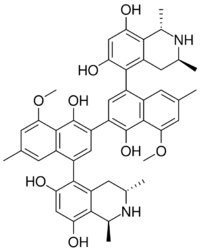
(1R,1′R,3R,3′R)-5,5′-(1,1′-Dihydroxy-8,8′-dimethoxy-6,6′-dimethyl[2,2′-binaphthalene]-4,4′-diyl)bis(1,3-dimethyl-1,2,3,4-tetrahydroisoquinoline-6,8-diol) | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C46H48N2O8 | |
| Molar mass | 756.896 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Michellamines are a group of atropisomeric alkaloid which have been found to be HIV viral replication inhibitors in vitro. It was discovered in the leaves of Ancistrocladus korupensis.[1] There are three michellamines represented as A, B, and C; however, michellamine B is the most active against the NID-DZ strain of HIV-2.[2]
Occurrence
Michellamine A and B occur naturally in Ancistrocladus korupensis leaves. Other chemical substances including alkaloids, tannins, and saponins are found in the roots, leaves, stems, flowers, or bark.[citation needed]
Synthesis
There are two methods explored to synthesize michellamines A and B. The first one, originally synthesized in 1994, is a retrosynthesis that leads to a biomimetic pathway that uses the construction of naphthalene/isoquinoline bonds before the naphthalene/naphthalene axis. The second method, originally synthesized only a few montes after the first method, is a complementary pathway that would use the naphthalene/naphthalene axis after it is created and add the two isoquinoline moieties.[3]
Research
Michellamines inhibit protein kinase C and virus-induced cellular fusion.[4] They have a broad range of effectiveness in vitro across most HIV strains, particularly the HIV-2 strain, which is found primarily in and around Cameroon.[4]
References
- ↑ Schlauer, Jan (1 February 1998). "Characterization of Enzymes fromAncistrocladus (Ancistrocladaceae) and Triphyophyllum (Dioncophyllaceae) Catalyzing Oxidative Coupling of Naphthylisoquinoline Alkaloids to Michellamines". Archives of Biochemistry and Biophysics 350 (1): 87–94. doi:10.1006/abbi.1997.0494. PMID 9466824.
- ↑ Zhang, Heping; Zembower, David; Chen, Zhidong (October 1997). "Structural analogues of the michellamine anti-HIV agents. Importance of the tetrahydroisoquinoline rings for biological activity". Bioorganic & Medicinal Chemistry Letters 7 (20): 2687–2690. doi:10.1016/S0960-894X(97)10057-9.
- ↑ Bringmann, Gerhard; Götz, Roland; Keller, Paul A.; Walter, Rainer; Boyd, Michael R.; Lang, Fengrui; Garcia, Alberto; Walsh, John J. et al. (January 1998). "A Convergent Total Synthesis of the Michellamines". The Journal of Organic Chemistry 63 (4): 1090–1097. doi:10.1021/jo971495m.
- ↑ 4.0 4.1 White, E.; Chao, W. R.; Ross, L. J.; Borhani, D. W.; Hobbs, P. D.; Upender, V.; Dawson, M. I. (1999). "Michellamine Alkaloids Inhibit Protein Kinase C". Archives of Biochemistry and Biophysics 365 (1): 25–30. doi:10.1006/abbi.1999.1145. PMID 10222035.
