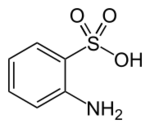
Chemistry:Orthanilic acid
From HandWiki
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Aminobenzene-1-sulfonic acid | |
| Other names
Orthanilic acid, 2-Aminobenzenesulfonic acid
o-Aminobenzenesulfonic acid Aniline-2-sulfonic acid 88-21-1 Aniline-o-sulfonic acid[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H7NO3S | |
| Molar mass | 173.19 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Orthanilic acid (2-aminobenzenesulfonic acid) is a biological acid with roles in benzoate degradation and microbial metabolism in diverse environments.
Orthanilic acid promotes reverse turn formation in peptides, inducing a folded conformation[2][3] when incorporated into peptide sequences (Xaa-SAnt-Yaa), showing robust 11-membered-ring hydrogen-bonding.
Orthanilic acid is a structural component of some azo dyes which consequently have poor bacterial degradation.[4]
Orthanilic acids have also been found to affect cardiac tension.[5][6]
References
- ↑ "PubChem entry". https://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=6926.
- ↑ Kale, Sangram S.; Priya, Gowri; Kotmale, Amol S.; Gawade, Rupesh L.; Puranik, Vedavati G.; Rajamohanan, P. R.; Sanjayan, Gangadhar J. (March 2013). "Orthanilic acid-promoted reverse turn formation in peptides". Chem. Commun. 49 (22): 2222–4. doi:10.1039/C3CC40522B. PMID 23392615.
- ↑ Kale, Sangram S.; Priya, Gowri; Kotmale, Amol S.; Gawade, Rupesh L.; Puranik, Vedavati G.; Rajamohanan, P. R.; Sanjayan, Gangadhar J. (2013). "Orthanilic acid-promoted reverse turn formation in peptides". Chemical Communications 49 (22): 2222–4. doi:10.1039/C3CC40522B. PMID 23392615.
- ↑ Tan, Nico C. G.; Leeuwen, Annemarie van; Voorthuizen, Ellen M. van; Slenders, Peter; Prenafeta-Boldú, Francesc X.; Temmink, Hardy; Lettinga, Gatze; Field, Jim A. (2005). "Fate and biodegradability of sulfonated aromatic amines". Biodegradation 16 (6): 527–37. doi:10.1007/s10532-004-6593-x. PMID 15865345.
- ↑ Franconi, F; Bennardini, F; Campana, S; Failli, P; Matucci, R; Stendardi, I; Giotti, A (1990). "Effect of taurine, L-cysteic and orthanilic acids on cardiac tension". Progress in Clinical and Biological Research 351: 175–84. PMID 2122477.
- ↑ Franconi, F; Bennardini, F; Campana, S; Failli, P; Matucci, R; Stendardi, I; Giotti, A (1990). "Effect of taurine, L-cysteic and orthanilic acids on cardiac tension". Prog. Clin. Biol. Res. 351: 175–84. PMID 2122477.
 |

