Chemistry:Oxidation with chromium(VI)-amine complexes
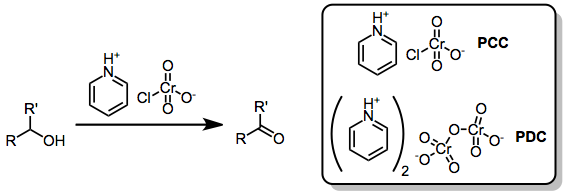
Oxidation with chromium(VI)-amine complexes involves the conversion of alcohols to carbonyl compounds or more highly oxidized products through the action of chromium(VI) oxide-amine adducts and salts. Representative members of this family of reagents include Collins reagent, pyridinium chlorochromate (PCC), and pyridinium dichromate (PDC).[1]
Introduction
Sarrett identified the adduct of pyridine and chromium(VI) oxide (Collins reagent) as a selective compound for the oxidation of primary and secondary alcohols to carbonyl compounds.[2] Despite its selectivity, Collins reagent suffers from difficulties associated with its preparation, stability, and efficiency. The less reactive adducts pyridinium chlorochromate (PCC) and pyridinium dichromate (PDC) are more easily handled and more selective than Collins reagent in oxidations of alcohols. These reagents, as well as other, more exotic adducts of nitrogen heterocycles with chromium(VI), facilitate a number of oxidative transformations of organic compounds, including cyclization to form tetrahydrofuran derivatives and allylic transposition to afford enones from allylic alcohols.
Oxidation with chromium(VI) amines has two primary limitations. Operationally, the tarry byproducts of chromium oxidations cause reduced yields and product sequestration.[3] In addition, Cr(VI)-amines (particularly PCC) may react with acid-labile functionality. Thus, these agents have been employed in oxidations of relatively simple substrates, often in excess to account for reagent trapping and decomposition. The use of adsorbents such as Celite or silica gel facilitates the removal of chromium byproducts and eliminates many of the operational difficulties associated with chromium-mediated oxidations.
(1)
Mechanism and stereochemistry
Prevailing mechanisms
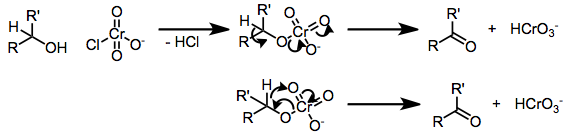
Chromate esters have been implicated in most oxidations of alcohols by chromium(VI)-amines. Upon formation of the chromate ester, either deprotonation or hydride transfer leads to the product carbonyl compound. Kinetic isotope effect studies have shown that C-H bond cleavage is involved in the rate-determining step.[4]
(2)
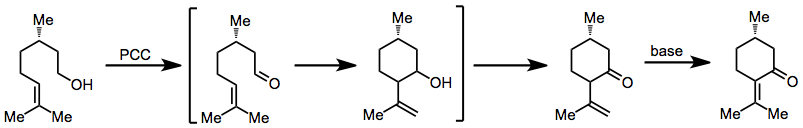
Oxidative annulation of alkenols to form six-membered rings may be accomplished with PCC. This process is postulated to occur via initial oxidation of the alcohol, attack of the alkene on the new carbonyl, then re-oxidation to a ketone. Double-bond isomerization may occur upon treatment with base as shown in equation (3) below.[5]
(3)
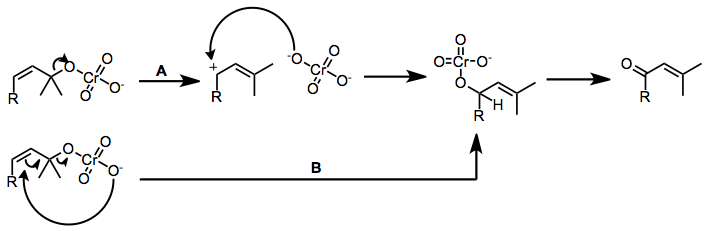
An important process mediated by chromium(VI)-amines is the oxidative transposition of tertiary allylic alcohols to give enones.[6] The mechanism of this process likely depends on the acidity of the chromium reagent. Acidic reagents such as PCC may cause ionization and recombination of the chromate ester (path A), while the basic reagents (Collins) likely undergo direct allylic transposition via sigmatropic rearrangement (path B).
(4)
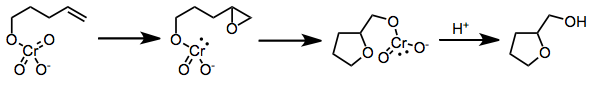
Oxidative cyclizations of olefinic alcohols to cyclic ethers may occur via [3+2], [2+2],[7] or epoxidation mechanisms. The exact mechanism has been debated, although a recent structure-reactivity study provided evidence for direct epoxidation by the chromate ester.[8] Subsequent epoxide opening and release of chromium leads to the observed products.
(5)
Scope and limitations
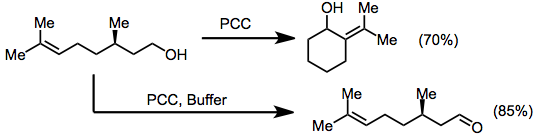
Buffering agents may be used to prevent acid-labile protecting groups from being removed during chromium(VI)-amine oxidations. However, buffers will also slow down oxidative cyclizations, leading to selective oxidation of alcohols over any other sort of oxidative transformation. Citronellol, for instance, which cyclizes to pugellols in the presence of PCC, does not undergo cyclization when buffers are used.[9][10]
(6)
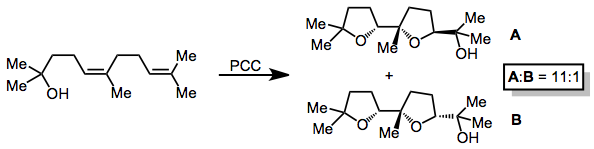
Oxidative cyclization can be used to prepare substituted tetrahydrofurans. Cyclization of dienols leads to the formation of two tetrahydrofuran rings in a syn fashion.[11]
(7)
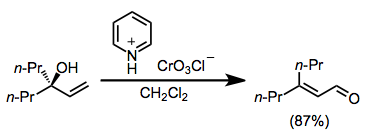
Enones can be synthesized from tertiary allylic alcohols through the action of a variety of chromium(VI)-amine reagents. The reaction is driven by the formation of a more substituted double bond. (E)-Enones form in greater amounts than (Z) isomers because of chromium-mediated geometric isomerization.[10][12]
(8)
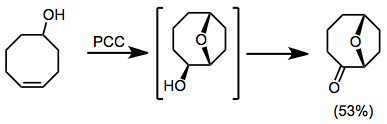
Suitably substituted olefinic alcohols undergo oxidative cyclization to give tetrahydrofurans. Further oxidation of these compounds to give tetrahydropyranyl carbonyl compounds then occurs.[13]
(9)
In addition to the limitations described above, chromium(VI) reagents are often unsuccessful in the oxidation of substrates containing heteroatoms (particularly nitrogen). Coordination of the heteroatoms to chromium (with displacements of the amine ligand originally attached to the metal) leads to deactivation and eventual decomposition of the oxidizing agent.
Comparison with other methods
Methods employing dimethyl sulfoxide (the Swern and Moffatt oxidations) are superior to chromium(VI)-amines for oxidations of substrates with heteroatom functionality that may coordinate to chromium.[14] Dess-Martin periodinane (DMP) offers the advantages of operational simplicity, a lack of heavy metal byproducts, and selective oxidation of complex, late-stage synthetic intermediates.[15] Additionally, both DMP and manganese dioxide (MnO2) can be used to oxidize allylic alcohols to the corresponding enones without allylic transposition. When allylic transpositions is desired, however, chromium(VI)-amine reagents are unrivaled.
Catalytic methods employing cheap, clean terminal oxidants in conjunction with catalytic amounts of chromium reagents produce only small amounts of metal byproducts.[16] However, undesired side reactions mediated by stoichiometric amounts of the terminal oxidant may occur.
References
- ↑ Luzzio, F. A. Org. React. 1998, 53, 1. doi:10.1002/0471264180.or053.01
- ↑ Poos, G. I.; Arth, G. E.; Beyler, R. E.; Sarrett, L. H. J. Am. Chem. Soc., 1953, 75, 422.
- ↑ Ratcliffe, R.; Rodehorst, R. J. Org. Chem., 1970, 35, 4000.
- ↑ Banerji, K. K. J. Org. Chem., 1988, 53, 2154.
- ↑ Corey, E. J.; Boger, D. Tetrahedron Lett., 1978, 19, 2461.
- ↑ Luzzio, F. A.; Moore, W. J. J. Org. Chem., 1993, 58, 2966.
- ↑ Piccialli, V. Synthesis 2007, 2585.
- ↑ Beihoffer, L.A; Craven, R.A.; Knight, K.S; Cisson, C.R.; Waddell, T.G. Trans. Met. Chem. 2005, 30, 582.
- ↑ Fieser, L. F.; Fieser, M. Reagents for Organic Synthesis; Wiley-Interscience, New York, 1979, 7, 309.
- ↑ 10.0 10.1 Babler, J. H.; Coghlan, M. J. Synth. Commun. 1976, 6, 469.
- ↑ McDonald, F. E.; Towne, T. B. J. Am. Chem. Soc., 1994, 116, 7921.
- ↑ Majetich, G.; Condon, S.; Hull, K.; Ahmad, S. Tetrahedron Lett., 1989, 30, 1033.
- ↑ Schlecht, M. F.; Kim, H.-J. Tetrahedron Lett., 1986, 27, 4889.
- ↑ Tidwell, T. Org. React. 1990, 39, 297.
- ↑ Dess, D. B.; Martin, J. C. J. Org. Chem., 1983, 48, 4156.
- ↑ Muzart, J. Tetrahedron Lett., 1987, 28, 2133.
