Chemistry:Phosphine oxides
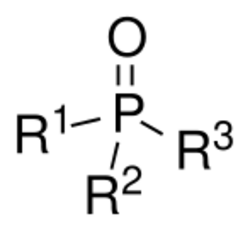
Phosphine oxides are phosphorus compounds with the formula OPX3. When X = alkyl or aryl, these are organophosphine oxides. Triphenylphosphine oxide is an example. An inorganic phosphine oxide is phosphoryl chloride (POCl3).[1] The parent phosphine oxide (H3PO) remains rare and obscure.
Structure and bonding
Tertiary phosphine oxides
Tertiary phosphine oxides are the most commonly encountered phosphine oxides. With the formula R3PO, they are tetrahedral compounds. They are usually prepared by oxidation of tertiary phosphines. The P-O bond is short and polar. According to molecular orbital theory, the short P–O bond is attributed to the donation of the lone pair electrons from oxygen p-orbitals to the antibonding phosphorus-carbon bonds.[2] The nature of the P–O bond was once hotly debated. Some discussions invoked a role for phosphorus-centered d-orbitals in bonding, but this analysis is not supported by computational analyses. In terms of simple Lewis structure, the bond is more accurately represented as a dative bond, as is currently used to depict an amine oxide.[3][4]
Secondary phosphine oxides
Secondary phosphine oxides (SPOs), formally derived from secondary phosphines (R2PH), are again tetrahedral at phosphorus.[5] One commercially available example of a secondary phosphine oxide is diphenylphosphine oxide. SPOs are used in the formulation of catalysts for cross coupling reactions.[6]
Unlike tertiary phosphine oxides, SPOs often undergo further oxidation, which enriches their chemistry:
- R2P(O)H + H2O2 → R2P(O)OH + H2O
These reactions are preceded by tautomerization to the phosphinous acid (R2POH):
- R
2P(O)H → R
2POH
Primary phosphine oxides
Primary phosphine oxides, formally oxidized derivatives of primary phosphines, are again tetrahedral at phosphorus. With four different substituents (O, OH, H, R) they are chiral. The primary phosphine oxides subject to tautomerization, which leads to racemization, and further oxidation, analogous to the behavior of SPOs. Additionally, primary phosphine oxides are susceptible to disproportionation to the phosphinic acid and the primary phosphine:[7]
- 2 RP(O)H
2 → RP(O)(H)OH + RPH
2 - 2 RP(O)H2 → RP(O)(H)OH + 2 RPH2
Syntheses
Phosphine oxide are typically produced by oxidation of organophosphines. The oxygen in air is often sufficiently oxidizing to fully convert trialkylphosphines to their oxides at room temperature:
- R3P + 1/2 O2 → R3PO
This conversion is usually undesirable. In order to suppress this reaction, air-free techniques are often employed when handling say, trimethylphosphine.
Less basic phosphines, such as methyldiphenylphosphine are converted to their oxides with hydrogen peroxide:[8]
- PMePh2 + H2O2 → OPMePh2 + H2O
Phosphine oxides are generated as a by-product of the Wittig reaction:
- R3PCR'2 + R"2CO → R3PO + R'2C=CR"2
Another albeit unconventional route to phosphine oxides is the thermolysis of phosphonium hydroxides:
- [PPh4]Cl + NaOH → Ph3PO + NaCl + PhH
The hydrolysis of phosphorus(V) dihalides also affords the oxide:[9]
- R3PCl2 + H2O → R3PO + 2 HCl
A special nonoxidative route is applicable secondary phosphine oxides, which arise by the hydrolysis of the chlorophosphine. An example is the hydrolysis of chlorodiphenylphosphine to give diphenylphosphine oxide:
- Ph2PCl + H2O → Ph2P(O)H + HCl
Reactions
Transition metal complexes of phosphine oxides are numerous.
Some phosphine oxides are well-known photoinitiators in photopolymer chemistry. UV/LED exposure induces a type I Norrish fission to free radicals, which then polymerize in a radical chain. An example is 2,4,6‑trimethylbenzoyldiphenylphosphine oxide, which absorbs around 380-410nm (near UV).[10]
Deoxygenation
Phosphine oxide deoxygenation has been extensively developed because many useful reactions convert stoichiometric tertiary phosphines to the corresponding oxides. Regenerating the tertiary phosphines requires cheap oxophilic reagents,[11] and can retain or invert chirality at P, depending on the reductant.[12]
Industrial deoxygenation usually occurs in two steps. Phosgene or equivalents first produce chlorotriphenylphosphonium chloride, which is then reduced separately.[13]
In the laboratory, phosphine oxides are usually reduced with silicon derivatives,[11] typically inexpensive trichlorosilane.
Trichlorosilane and triethylamine reduce phosphine oxides with inversion, whereas the reaction proceeds with retention absent the base:Cite error: Closing </ref> missing for <ref> tag
Use
Phosphine oxides are ligands in various applications of homogeneous catalysis. In coordination chemistry, they are known to have labilizing effects to CO ligands cis to it in organometallic reactions. The cis effect describes this process.
References
- ↑ D. E. C. Corbridge "Phosphorus: An Outline of its Chemistry, Biochemistry, and Technology" 5th Edition Elsevier: Amsterdam 1995. ISBN 0-444-89307-5.
- ↑ D. B. Chesnut (1999). "The Electron Localization Function (ELF) Description of the PO Bond in Phosphine Oxide". Journal of the American Chemical Society 121 (10): 2335–2336. doi:10.1021/ja984314m. Bibcode: 1999JAChS.121.2335C.
- ↑ Gilheany, Declan G. (1994). "No d Orbitals but Walsh Diagrams and Maybe Banana Bonds: Chemical Bonding in Phosphines, Phosphine Oxides, and Phosphonium Ylides". Chemical Reviews 94 (5): 1339–1374. doi:10.1021/cr00029a008. PMID 27704785.
- ↑ In fact, the N-O bonds in amine oxides are more likely to be closer to double bonds than are those of the P-O bonds in phosphine oxides; see e.g. https://pubs.rsc.org/en/content/articlelanding/2015/sc/c5sc02076j#:~:text=Quantitative%20analysis%20of%20known%20species%20of%20general%20formulae,high%20degree%20of%20covalent%20rather%20than%20ionic%20bonding.
- ↑ Gallen, Albert; Riera, Antoni; Verdaguer, Xavier; Grabulosa, Arnald (2019). "Coordination Chemistry and Catalysis with Secondary Phosphine oxides". Catalysis Science & Technology 9 (20): 5504–5561. doi:10.1039/C9CY01501A.
- ↑ Ackermann, Lutz (2007). "Catalytic Arylations with Challenging Substrates: From Air-Stable HASPO Preligands to Indole Syntheses and C-H-Bond Functionalizations". Synlett 2007 (4): 0507–0526. doi:10.1055/s-2007-970744.
- ↑ Horký, Filip; Císařová, Ivana; Štěpnička, Petr (2021). "A Stable Primary Phosphane Oxide and Its Heavier Congeners". Chemistry – A European Journal 27 (4): 1282–1285. doi:10.1002/chem.202003702. PMID 32846012.
- ↑ Denniston, Michael L.; Martin, Donald R. (1977). "Methyldiphenylphosphine Oxide and Dimethylphenylphosphine Oxide". Inorganic Syntheses. 17. pp. 183–185. doi:10.1002/9780470132487.ch50. ISBN 9780470132487.
- ↑ W. B. McCormack (1973). "3-Methyl-1-Phenylphospholene oxide". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=CV5P0787.; Collective Volume, 5, pp. 787
- ↑ "Boosting the cure of phosphine oxide photoinitiators" (in en). https://www.radtech.org/proceedings/2016/papers/technical-conference/Photoinitiator/Boosting%20the%20cure%20of%20phosphine%20oxide%20photoinitiators.pdf.
- ↑ 11.0 11.1 Podyacheva, Evgeniya; Kuchuk, Ekaterina; Chusov, Denis (2019). "Reduction of phosphine oxides to phosphines". Tetrahedron Letters 60 (8): 575–582. doi:10.1016/j.tetlet.2018.12.070.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedStereo - ↑ van Kalkeren, Henri A.; van Delft, Floris L.; Rutjes, Floris P. J. T. (2013). "Organophosphorus Catalysis to Bypass Phosphine Oxide Waste". ChemSusChem 6 (9): 1615–1624. doi:10.1002/cssc.201300368. ISSN 1864-5631. PMID 24039197. Bibcode: 2013ChSCh...6.1615V.
 |

