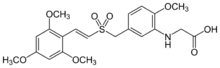
Chemistry:Rigosertib
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
(2-Methoxy-5-{[(E)-2-(2,4,6-trimethoxyphenyl)ethene-1-sulfonyl]methyl}phenyl)glycine
| |
| Systematic IUPAC name
(2-Methoxy-5-{[(E)-2-(2,4,6-trimethoxyphenyl)ethene-1-sulfonyl]methyl}anilino)acetate | |
| Other names
ON-01910
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| Properties | |
| C21H25NO8S | |
| Molar mass | 451.49 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Rigosertib (ON-01910 sodium salt, with Estybon as trade name) is a synthetic benzyl styryl sulfone in development by Onconova Therapeutics.[2] Rigosertib is in phase III clinical trials for the treatment of chronic myelomonocytic leukemia.[3]
Its geometrical isomer (Z)-ON 01910·Na has less cytotoxicity on cancer cells.
Mechanism
Rigosertib is a microtubule-destabilizing agent.[4]
References
- ↑ "physical and chemical data on chemispider website". http://www.chemspider.com/Chemical-Structure.5293927.html?rid=536d329b-927e-45a8-8495-63b421e6eba5.
- ↑ "Onconova Therapeutics". https://finpedia.co/bin/Companies/Onconova%20Therapeutics/.
- ↑ "Phase IIIB, Open-label, Multi-Center Study of the Efficacy and Safety of Rigosertib Administered as 72-hour Continuous Intravenous Infusions in Patients with Myelodysplastic Syndrome with Excess Blasts Progressing on or After Azacitidine or Decitabine". 29 June 2020. https://clinicaltrials.gov/ct2/show/NCT01928537.
- ↑ Jost, M (2017). "Combined CRISPRi/a-Based Chemical Genetic Screens Reveal that Rigosertib Is a Microtubule-Destabilizing Agent". Molecular Cell 68 (1): 210–223.e6. doi:10.1016/j.molcel.2017.09.012. PMID 28985505.
 |
