Chemistry:Saudin
From HandWiki
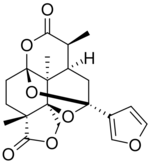
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
PubChem CID
|
|
| UNII | |
| |
| Properties | |
| C20H22O7 | |
| Molar mass | 374.389 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Saudin is a diterpenoid first isolated from the African flowering plant Cluytia richardiana.[1]
Saudin has shown a hypoglycemic effect in an rodent model experiment.[2]
Because of the unusual chemical structure and its potential biological activity, there has been research aimed at its total synthesis.[3][4][5][6][7]
References
- ↑ Mossa, Jaber S.; Cassady, John M.; Antoun, Mikhail D.; Byrn, Stephen R.; McKenzie, Ann T.; Kozlowski, John F.; Main, P. (1985). "Saudin, a hypoglycemic diterpenoid, with a novel 6,7-seco-labdane carbon skeleton, from Cluytia richardiana". The Journal of Organic Chemistry 50 (6): 916–918. doi:10.1021/jo00206a050.
- ↑ Mossa, J. S.; El-Denshary, E. S. M.; Hindawi, R.; Ageel, A. M. (1988). "The Hypoglycaemic Effect of Saudin". International Journal of Crude Drug Research 26 (2): 81–87. doi:10.3109/13880208809053897.
- ↑ Boeckman, Robert K.; Rosario Ferreira, Maria Rico del; Mitchell, Lorna H.; Shao, Pengcheng; Neeb, Michael J.; Fang, Yue (2011). "Studies culminating in the total synthesis and determination of the absolute configuration of (−)-saudin". Tetrahedron 67 (51): 9787–9808. doi:10.1016/j.tet.2011.09.067. PMID 22523435.
- ↑ Labadie, Guillermo R.; Cravero, Raquel M.; González-Sierra, Manuel (1996). "Studies Toward the Total Synthesis of Saudine : Simple and Stereoselective Synthesis of a Model Caged Ketal Backbone". Synthetic Communications 26 (24): 4671–4684. doi:10.1080/00397919608004793.
- ↑ Boeckman, Robert K.; Del Rosario Rico Ferreira, Maria; Mitchell, Lorna H.; Shao, Pengcheng (2002). "An Enantioselective Total Synthesis of (+)- and (−)-Saudin. Determination of the Absolute Configuration". Journal of the American Chemical Society 124 (2): 190–191. doi:10.1021/ja017194i. PMID 11782168.
- ↑ Tambar, Uttam K.; Kano, Taichi; Stoltz, Brian M. (2005). "Progress toward the Total Synthesis of Saudin: Development of a Tandem Stille-Oxa-Electrocyclization Reaction". Organic Letters 7 (12): 2413–2416. doi:10.1021/ol050705b. PMID 15932211. https://authors.library.caltech.edu/74471/2/ol050705bsi20050421_081713.pdf.
- ↑ Tambar, U. K.; Kano, T.; Zepernick, J. F.; Stoltz, B. M. (2008). "Convergent and diastereoselective synthesis of the polycyclic pyran core of saudin". The Journal of Organic Chemistry 71 (22): 8357–64. doi:10.1021/jo061236+. PMID 17064005. https://authors.library.caltech.edu/74508/5/jo061236%252Bsi20060901_075757.pdf.
 |
