Chemistry:Tantalate
From HandWiki
Short description: Group of chemical compounds
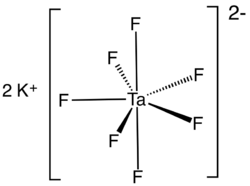
A tantalate is a tantalum-containing anion or a salt of such an anion. A commercially important example is heptafluorotantalate (TaF72−) and its potassium salt (K2TaF7).
Many oxides of tantalum are called tantalates. They are viewed as derivatives of "tantalic acid", hypothetic compounds with the formulas Ta2O5·nH2O[1] or HTaO3[2]). Examples of such tantalates are lithium tantalate (LiTaO3), lutetium tantalate (LuTaO4), neodymium tantalate (NdTaO4) and lead scandium tantalate (PST or Pb(ScxTa1-x)O3. Polyoxometallates containing tantalum provide examples of discrete tantalum oxides that exist in solution.
References
- ↑ Szanics, Judit; Kakihana, Masato (1999). "A Novel Tantalic Acid-Based Polymerizable Complex Route to LiTaO3 Using Neither Alkoxides nor Chlorides of Tantalum". Chemistry of Materials 11 (10): 2760. doi:10.1021/cm990160d.
- ↑ Inoue, Y (1996). "Synthetic inorganic ion exchange materials XLI: Ion exchange properties of cubic tantalic acid (HTaO3)". Materials Research Bulletin 31 (6): 691–698. doi:10.1016/0025-5408(96)00050-5.
 |