Chemistry:Tetramethylethylenediamine(dimethyl)nickel(II)
From HandWiki
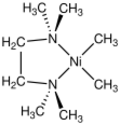
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| C8H22N2Ni | |
| Molar mass | 204.971 g·mol−1 |
| Appearance | yellow-brown solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Tetramethylethylenediamine(dimethyl)nickel(II) is the organonickel complex with the formula (Me
2NCH
2CH
2NMe
2)NiMe
2 (Me = CH3). This yellow-brown, air-sensitive compound is popular precursor to diverse organonickel complexes. It is prepared from the tmeda adduct of nickel(II) acetylacetonate by reaction with methyl lithium.[1][2]
The tmeda ligand is easily displaced by bases such as bipyridine and diphosphines. Treatment of the complex with electrophilic alkenes results in elimination of ethylene, giving alkene complexes.[2]
References
- ↑ Göttker-Schnetmann, Inigo; Mecking, Stefan (2020). "A Practical Synthesis of (tmeda)Ni(CH3)2, Isotopically Labeled (tmeda)Ni(13CH3)2, and Neutral Chelated-Nickel Methyl Complexes". Organometallics 39 (18): 3433–3440. doi:10.1021/acs.organomet.0c00500.
- ↑ 2.0 2.1 Kaschube, Wilfried; Pörschke, Klaus R.; Wilke, Günther (1988). "Tmeda-Nickel-Komplexe". Journal of Organometallic Chemistry 355 (1–3): 525–532. doi:10.1016/0022-328X(88)89050-8.
 |

