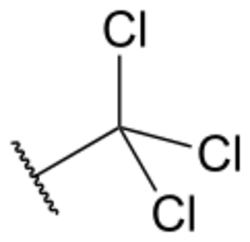
Chemistry:Trichloromethyl group
The trichloromethyl group is a functional group that has the formula –CCl3. The naming of is group is derived from the methyl group (which has the formula –CH3), by replacing each hydrogen atom by a chlorine atom. Compounds with this group are a subclass of the organochlorines. Some notable examples of compounds with this group are trichloromethane H–CCl3, 1,1,1-trichloroethane H3C–CCl3, and chloral HOC–CCl3.
The trichloromethyl group has a significant electronegativity.[citation needed] For this reason, trichloromethyl-substituted acids, such as trichloromethanesulfonic acid, are often stronger than the original. For example, the acidity constant (pKa) of trichloroacetic acid HOOC–CCl3 is 0.77, whereas that of acetic acid is 4.76.[1][2]
By the same principle, the trichloromethyl group generally lowers the basicity of organic compounds, e.g. trichloroethanol.
See also
- Trifluoromethyl group
- Trichloromethoxy
References
 |