Earth:Peroxyacyl nitrates
In organic chemistry, peroxyacyl nitrates (also known as Acyl peroxy nitrates, APN or PANs) are powerful respiratory and eye irritants present in photochemical smog. They are nitrates produced in the thermal equilibrium between organic peroxy radicals by the gas-phase oxidation of a variety of volatile organic compounds (VOCs). Another way to produce PANs is by aldehydes and other oxygenated VOCs oxidizing in the presence of NO2.[1][2][3]
They are good markers for the source of VOCs as either biogenic or anthropogenic, which is useful in the study of global and local effects of pollutants.[4][5]
Formation
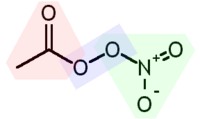
PANs are secondary pollutants, which means they are not directly emitted as exhaust from power plants or internal combustion engines, but they are formed from other pollutants by chemical reactions in the atmosphere. Free radical reactions catalyzed by ultraviolet light from the sun oxidize unburned non-methane[6] hydrocarbons to aldehydes, ketones, and dicarbonyls, whose secondary reactions create peroxyacyl radicals. The most common peroxyacyl radical is peroxyacetyl, which can be formed from the free radical oxidation of acetaldehyde, various ketones, or the photolysis of dicarbonyl compounds such as methylglyoxal or diacetyl.
- Hydrocarbons + O2 + light → RC(O)OO•
These react reversibly with nitrogen dioxide (NO2) to form PANs:[6]
- RC(O)OO• + NO2• ⇌ RC(O)OONO2
Night-time reaction of aldehydes with nitrogen trioxide is another possible source.[6]
The stability of PANs in the atmosphere is dependent on temperature. The lower temperatures in the troposphere increase the stability and therefore lifetime of PANs.[7] Since they dissociate quite slowly in the atmosphere into radicals and NO2, PANs are able to transport these unstable compounds far away from their urban and industrial origins. This causes a decrease in photochemical ozone production near sources of NOx as PANs are a NOx reservoir, reducing the amount of NOx photolyzed.[8] This allows PANs to transport NOx to regions where it can more efficiently produce tropospheric ozone.
Types
Peroxyacetyl nitrate is the first member of PANs identified by scientists in the 1950s and the most prevalent peroxyacyl nitrate (75–90% of total atmospheric emissions),[6][9] followed by peroxypropionyl nitrate (PPN),[10] the second member of PANs discovered from synthetic mixtures.[9] The third member of the PANs class is peroxybutyryl nitrate (PBN), which is only been known to be synthetically made.[9] Peroxybenzoyl nitrate (PBzN)[10] and methacryloyl peroxynitrate (MPAN)[4] have also been observed. The composition of PANs in a particular region depends heavily on which hydrocarbons are present in the atmosphere, with the exception of peroxyacetyl nitrate, which is able to be produced from a range of precursors.[4]: 7624
Health effects
PANs are both toxic and irritating, as they dissolve more readily in water than ozone. They are lachrymators, causing eye irritation at concentrations of only a few parts per billion. At higher concentrations they cause extensive damage to vegetation.[11]

References
- ↑ Cape, J.N. (2003). "Effects of airborne volatile organic compounds on plants". Environmental Pollution 122 (1): 145–157. doi:10.1016/S0269-7491(02)00273-7. PMID 12535603. Bibcode: 2003EPoll.122..145C.
- ↑ Gaffney, Jeffrey S.; Marley, Nancy A. (2021). "The Impacts of Peroxyacetyl Nitrate in the Atmosphere of Megacities and Large Urban Areas: A Historical Perspective". ACS Earth and Space Chemistry 5 (8): 1829–1841. doi:10.1021/acsearthspacechem.1c00143. Bibcode: 2021ESC.....5.1829G.
- ↑ Jickells, T.; Baker, A.R.; Cape, J.N.; Cornell, S.E.; Nemitz, E. (2013). "The cycling of organic nitrogen through the atmosphere". Philosophical Transactions of the Royal Society B: Biological Sciences 368 (1621). doi:10.1098/rstb.2013.0115.
- ↑ 4.0 4.1 4.2 LaFranchi, B.W.; Wolfe, G.M. (2009). "Closing the peroxy acetyl nitrate budget: observations of acyl peroxy nitrates (PAN, PPN, and MPAN) during BEARPEX 200". Atmos. Chem. Phys. 9 (19): 7623–7641. doi:10.5194/acp-9-7623-2009. Bibcode: 2009ACP.....9.7623L.
- ↑ Thornton, Joel. "PANs". Department of Atmospheric Sciences, University of Washington.
- ↑ 6.0 6.1 6.2 6.3 Fischer, E.V.; Jacob, D.J.; Yantosca, R.M.; Sulprizio, M.P.; Millet, D.B.; Mao, J.; Paulot, F.; Singh, H.B. et al. (14 March 2014). "Atmospheric peroxyacetyl nitrate (PAN): a global budget and source attribution". Atmospheric Chemistry and Physics 14 (5): 2679–2698. doi:10.5194/acp-14-2679-2014. PMID 33758588. Bibcode: 2014ACP....14.2679F.
- ↑ Jenkin, Michael E.; Clemitshaw, Kevin C. (2000). "Ozone and other secondary photochemical pollutants: chemical processes governing their formation in the planetary boundary layer". Atmospheric Environment 34 (16): 2499–2527. doi:10.1016/S1352-2310(99)00478-1. Bibcode: 2000AtmEn..34.2499J.
- ↑ Gil, Junsu; Lee, Meehye; Han, Jihyun; Kim, Joo-Ae; Kim, Saewung; Guenther, Alex; Kim, Hyunseok; Kim, Soyoung et al. (2018). "Peroxyacetyl Nitrate and Ozone Enhancement at Taehwa Research Forest under the Influence of Seoul Metropolitan Area". Aerosol Air Qual. Res. 18 (9): 2262–2273. doi:10.4209/aaqr.2017.11.0451. Bibcode: 2018AAQR...18.2262G.
- ↑ 9.0 9.1 9.2 Stephens, E.R.; Burleson, F.R.; Cardiff, E.A. (1965). "The Production of Pure Peroxyacyl Nitrates". Journal of the Air Pollution Control Association 15 (3): 87–89. doi:10.1080/00022470.1965.10468346. PMID 14257516.
- ↑ 10.0 10.1 Kleindienst, Tadeusz E.; Shepson, Paul B.; Smith, David F.; Hudgens, Edward E.; Nero, Chris M.; Cupitt, Larry T.; Bufalini, Joseph J.; Claxton, Larry D. et al. (January 1990). "Comparison of mutagenic activities of several peroxyacyl nitrates". Environmental and Molecular Mutagenesis 16 (2): 70–80. doi:10.1002/em.2850160204. PMID 2209566. Bibcode: 1990EnvMM..16...70K.
- ↑ Mudd, J.B.; Kozlowski, T.T. (1975). Responses of Plants to Air Pollution. 111 Fifth Avenue, New York, New York 10003: Academic Press, INC.. pp. 97–116. ISBN 0-12-509450-7.
 |
