Physics:Evaporation suppressing monolayers
A major contributor to this article appears to have a close connection with its subject. (March 2014) (Learn how and when to remove this template message) |
Evaporation suppressing monolayers are materials that when applied to the air/water interface, will spread (or self-assemble) and form a thin film across the surface of the water. The purpose of these materials is to reduce evaporative water loss from dams and reservoirs. File:Self Assembly of Octadecanol monolayer on water from random start.ogv
Mechanism
The specific mechanism that underlies monolayer evaporation resistance has been attributed to the physical barrier formed by the presence of these materials on the surface of the water (see figure).[1] The extent to which a material can resist the evaporation of water is best treated on a case-by-case basis, however, a number of empirically derived formulas have been reported. Before the advent of surface spectroscopic techniques such as Brewster Angle Microscopy (BAM) and Glancing Incidence X-Ray reflectometry (GIXD), it was thought that the intermolecular spacing between monolayer molecules was the largest determinant factor governing evaporation suppression. When the surface density of the monolayer was sufficiently small, water molecules were presumed to pass through the space between molecules.[2][3][4][5] Scattering and imaging results, however, revealed that the intermolecular distance between monolayer molecules was essentially constant, and that evaporation was more likely at domain boundaries.[6] The factors governing the efficacy of an evaporation suppressing monolayer are the ability to remain tightly packed despite changes in surface pressure, the ability to adhere to the surface of water, and to neighbouring molecules.[7]
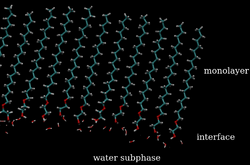
History
Irving Langmuir accurately described the geometric structure of a monolayer film on water in 1917, work for which he would be later awarded the Nobel prize in chemistry.[8] The evaporation suppressing properties of these materials were first reported by Rideal in the 1920s[9] In the 1940s Langmuir and Schaefer quantified the evaporation resistance and its dependence on temperature.[10] This work was extended by Archer and La Mer in the following decade, who observed a dependence on surface pressure, chain length, monolayer phase, subphase composition and surface temperature.[4] Large scale field trials were being conducted at this time in Australia by Mansfield[11] He reported that the results seen in the laboratory setting could not be replicated in real world conditions, with dust and wind being cited as adversely affecting evaporation suppressing performance.
In later decades, research efforts focussed on multicomponent monolayer materials such as hexadecanol + octadecanol,[12] altering the number of carbons in the aliphatic chain,[13] and later on, the addition of polymerised surfactants to increase monolayer stability.[14]
Better monolayer materials are required as are better methods of monolayer distribution methods, although no resolution of these difficulties presently exists.[15]
Despite research in this area for most of the 20th century, no durable, effective and inexpensive product has come to market. Recently, advances in experimental and modelling techniques have increased the understanding of these materials.
Recent Developments
During a prolonged drought in Australia at the start of this century, scientists there from a number of research institutions, including Pr. David Solomon, inventor of the polymer banknote,[16] set about developing a product that is efficacious, resistant to the deleterious effects of wind, and affordable. In addition to small and large scale field trials, new techniques were utilised including a novel evaporation tank with a controlled climate system to mimic the effects of wind and waves,[17] and computational modelling was for the first time employed to relate dynamic and geometric properties at the atomistic level, with evaporation suppressing performance at the macroscopic level.[18][19][20] The use of ethylene glycol monooctadecyl ether was found to substantially decrease evaporation resistance in the presence of wind, and the addition of a water-soluble polymer further enhanced its effectiveness.[7][21]
Commercial Products
WaterGuard manufactured by Aquatain advertises a polymer based material that reduces water evaporation.[22] Other products include Solarpill[23] and Water$aver.[24] The efficacy of these products has not been shown.
See also
- Self-assembled monolayer
- Monolayers
References
- ↑ Barnes, G., & Gentle, I. (2011). Interfacial science: an introduction. Oxford University Press.
- ↑ Blank, M. (1964). An approach to a theory of monolayer permeation by gases. The Journal of Physical Chemistry, 68(10), 2793-2800.
- ↑ Blank, M., & Britten, J. S. (1965) Transport properties of condensed monolayers. Journal of colloid science, 20(8), 789-800.
- ↑ 4.0 4.1 Archer, R. J., & Mer, V. K. L. (1955). The rate of evaporation of water through fatty acid monolayers. The Journal of Physical Chemistry, 59(3), 200-208.
- ↑ Barnes, G. T., Quickenden, T. I., & Saylor, J. E. (1970). A statistical calculation of monolayer permeation by water. Journal of Colloid and Interface Science, 33(2), 236-243.
- ↑ McNamee, C. E., Barnes, G. T., Gentle, I., Peng, J. B., Steitz, R., & Probert, R. (1998). The evaporation resistance of mixed monolayers of octadecanol and cholesterol. Journal of colloid and interface science, 207(2), 258-263.
- ↑ 7.0 7.1 Prime, E. L., Tran, D. N., Plazzer, M., Sunartio, D., Leung, A. H., Yiapanis, G., ... & Solomon, D. H. (2012). Rational design of monolayers for improved water evaporation mitigation. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 415, 47-58.
- ↑ Langmuir, I. (1916). THE CONSTITUTION AND FUNDAMENTAL PROPERTIES OF SOLIDS AND LIQUIDS. PART I. SOLIDS. Journal of the American Chemical Society, 38(11), 2221-2295.
- ↑ Rideal, E. K. (1925). On the influence of thin surface films on the evaporation of water. The Journal of Physical Chemistry, 29(12), 1585-1588.
- ↑ Langmuir, I., & Schaefer, V. J. (1943). Rates of evaporation of water through compressed monolayers on water. Journal of the Franklin Institute, 235(2), 119-162.
- ↑ Mansfield, W. W. Influence of monolayers on the natural rate of evaporation of water. Nature, 175, 247.
- ↑ Foulds, E. L., & Dressler, R. G. (1968). Performance of monolayer blends of odd and even carbon chain alcohols in water evaporation suppression. Industrial & Engineering Chemistry Product Research and Development, 7(1), 75-79.
- ↑ Simko, A. J., & Dressler, R. G. (1969). Investigation of C20 to C25 Fatty Alcohols and Blends as Water Evaporation Retardants. Industrial & Engineering Chemistry Product Research and Development, 8(4), 446-450.
- ↑ Fukuda, K., Kato, T., Machida, S., & Shimizu, Y. (1979). Binary mixed monolayers of polyvinyl stearate and simple long-chain compounds at the air/water interface. Journal of Colloid and Interface Science, 68(1), 82-95.
- ↑ Barnes, G.T. (2008) Review: The potential for monolayers to reduce the evaporation from large water storages. Agricultural Water Management, 95, 339-353
- ↑ "Polymer banknotes - CSIROpedia". http://www.csiropedia.csiro.au/display/CSIROpedia/Polymer+banknotes#Polymerbanknotes-Howitallstarted.
- ↑ Schouten, P., Putland, S., Lemckert, C., Underhill, I., Solomon, D., Sunartio, D., ... & Qiao, G. (2012). Evaluation of an evaporation suppressing monolayer system in a controlled wave tank environment: A pilot investigation. Australian Journal of Water Resources, 16(1).
- ↑ Henry, D. J., Dewan, V. I., Prime, E. L., Qiao, G. G., Solomon, D. H., & Yarovsky, I. (2010). Monolayer structure and evaporation resistance: A molecular dynamics study of octadecanol on water. The Journal of Physical Chemistry B, 114(11), 3869-3878.
- ↑ Plazzer, M. B., Henry, D. J., Yiapanis, G., & Yarovsky, I. (2011). Comparative study of commonly used molecular dynamics force fields for modeling organic monolayers on water. The Journal of Physical Chemistry B, 115(14), 3964-3971.
- ↑ Tran, D. N., Prime, E. L., Plazzer, M., Leung, A. H., Yiapanis, G., Christofferson, A. J., ... & Solomon, D. H. (2013). Molecular Interactions behind the Synergistic Effect in Mixed Monolayers of 1-Octadecanol and Ethylene Glycol Monooctadecyl Ether. The Journal of Physical Chemistry B, 117(13), 3603-3612.
- ↑ "AU2009001684 METHOD FOR CONTROLLING WATER EVAPORATION". Patentscope.wipo.int. http://patentscope.wipo.int/search/en/WO2010071931. Retrieved 2014-03-08.
- ↑ "Liquid Innovations! - Home". Aquatain.com. http://www.aquatain.com/. Retrieved 2014-03-08.
- ↑ "SmartPool AP72 SolarPill Liquid Ball Solar Blanket Cover for Pools up to 30,000 Gallons". PoolSupplyWorld.com. http://www.poolsupplyworld.com/SmartPool-AP72-SolarPill-Liquid-Ball-Solar-Blanket-Cover-for-Pools-up-to-30000-Gallons/AP72.htm. Retrieved 2014-03-08.
- ↑ "Watersavr". Flexible Solutions. http://www.flexiblesolutions.com/products/watersavr/. Retrieved 2014-03-08.
 |
