Chemistry:Tetramethylazodicarboxamide: Difference between revisions
From HandWiki
(update) |
(No difference)
|
Latest revision as of 01:18, 6 February 2024
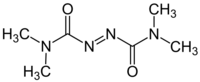
| |
| Names | |
|---|---|
| IUPAC name
3-(Dimethylcarbamoylimino)-1,1-dimethylurea
| |
| Other names
Diamide; Tetramethyldiazenedicarboxamide; N,N,N′,N′-Tetramethylazoformamide 1,1'-Azobis(N,N-dimethylformamide); N,N,N′,N′-Tetramethylazobisformamide; Azodicarboxylic acid bis(dimethylamide); 1,1'-Azobis(N,N-dimethylformamide)
| |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | TMAD |
| ChEBI | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C6H12N4O2 | |
| Molar mass | 172.188 g·mol−1 |
| Appearance | Yellow crystalline solid |
| Melting point | 113 to 115 °C (235 to 239 °F; 386 to 388 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Tetramethylazodicarboxamide (also known as TMAD and diamide) is a reagent used in biochemistry for oxidation of thiols in proteins to disulfides.[2] It has also been used as a reagent in the Mitsunobu reaction in place of diethyl azodicarboxylate.[1]
References
- ↑ 1.0 1.1 Tetsuto Tsunoda, Hiroto Kaku, N,N,N′,N′-Tetramethylazodicarboxamide, in Encyclopedia of Reagents for Organic Synthesis, 2003, John Wiley & Sons, Ltd, doi:10.1002/047084289X.rn00274
- ↑ CID 4278 from PubChem
Further reading
- Tsunoda, Tetsuto; Otsuka, Junko; Yamamiya, Yoshiko; Itô, Shô (1994). "N,N,N',N'-Tetramethylazodicarboxamide(TMAD), A New Versatile Reagent for Mitsunobu Reaction. Its Application to Synthesis of Secondary Amines". Chemistry Letters (3): 539. doi:10.1246/cl.1994.539.
 |

