Physics:Carbon quantum dots
| Part of a series of articles on |
| Nanomaterials |
|---|
 |
| Carbon nanotubes |
| Fullerenes |
| Other nanoparticles |
| Nanostructured materials |
|
|
Carbon quantum dots also commonly called Carbon dots (abbreviated as CQDs, C-dots or CDs) are carbon nanoparticles which are less than 10 nm in size and have some form of surface passivation.[1][2][3]
History
CQDs were first discovered by Xu et al. in 2004 accidentally during the purification of single-walled carbon nanotubes.[4] This discovery triggered extensive studies to exploit the fluorescence properties of CQDs.[1][5]
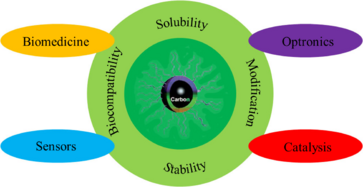
As a new class of fluorescent carbon nanomaterials, CQDs possess the attractive properties of high stability, good conductivity, low toxicity, environmental friendliness, simple synthetic routes as well as comparable optical properties to quantum dots.[6] Carbon quantum dots have been extensively investigated especially due to their strong and tunable fluorescence emission properties,[7] which enable their applications in biomedicine, optronics, catalysis, and sensing.[8]
The fundamental mechanisms responsible of the fluorescence capability of CQDs are very debated. Some authors have provided evidence of size-dependent fluorescence properties, suggesting that the emission arises from electronic transitions with the core of the dots, influenced by quantum confinement effects,[9][10] whereas other works have rather attributed the fluorescence to recombination of surface-trapped charges,[11][12] or proposed a form of coupling between core and surface electronic states.[13] The excitation-dependent fluorescence of CQDs, leading to their characteristic emission tunability, has been mostly linked to the inhomogeneous distribution of their emission characteristics,[14][13] due to polydispersity, although some works have explained it as a violation of Kasha's rule arising from an unusually slow solvent relaxation.[15]
Properties of CQDs
The structures and components of CQDs determine their diverse properties. Many carboxyl moieties on the CQD surface impart excellent solubility in water and biocompatibility.[7] Such surface moieties enable CQDs to serve as proton conducting nanoparticles.[16] CQDs are also suitable for chemical modification and surface passivation with various organic, polymeric, inorganic or biological materials. By surface passivation, the fluorescence properties as well as physical properties of CQDs are enhanced. Recently, it has been discovered that amine and hydroxamic acid functionalized CD can produce tricolor (green, yellow and red) emission when introduced with different pH environment and this tricolor emission can be preserved in ORMOSIL film matrix.[17] A paper published in 2019 showed that CQD can resist temperatures as high as 800 °C, paving way for applications of CQD in high temperature environments.[18] Based on carbon, CQDs possess such properties as good conductivity, benign chemical composition, photochemical and thermal stability.[citation needed]
Synthesis of CQDs
Synthetic methods for CQDs are roughly divided into two categories, "top-down" and "bottom-up" routes. These can be achieved via chemical, electrochemical or physical techniques.[7] The CQDs obtained could be optimized during preparation or post-treatment.[1] Modification of CQDs is also very important to get good surface properties which are essential for solubility and selected applications.[1]
Synthetic methods
"Top-down" synthetic route refers to breaking down larger carbon structures such as graphite, carbon nanotubes, and nanodiamonds into CQDs using laser ablation, arc discharge, and electrochemical techniques.[7] For example, Zhou et al. first applied electrochemical method into synthesis of CQDs.[19] They grew multi-walled carbon nanotubes on a carbon paper, then they inserted the carbon paper into an electrochemical cell containing supporting electrolyte including degassed acetonitrile and 0.1 M tetrabutyl ammonium perchlorate. Later, they applied this method in cutting CNTs or assembling CNTs into functional patterns which demonstrated the versatile callability of this method in carbon nanostructure manipulations.[20][21]
"Bottom-up" synthetic route involves synthesizing CQDs from small precursors such as carbohydrates, citrate, and polymer-silica nanocomposites through hydrothermal/solvothermal treatment, supported synthetic, and microwave synthetic routes.[22] For instance, Zhu et al. described a simple method of preparing CQDs by heating a solution of poly(ethylene glycol) (PEG) and saccharide in 500 W microwave oven for 2 to 10 min.[23]
Recently, green synthetic approaches have also been employed for fabrication of CQDs.[24][25][26][27][28]
Size control
In addition to post-treatment, controlling the size of CQDs during the preparing process is also widely used. For instance, Zhu et al. reported hydrophilic CQDs through impregnation of citric acid precursor.[23] After pyrolyzing CQDs at 300 °C for 2 hours in air, then removing silica, followed by dialysis, they prepared CQDs with a uniform size of 1.5–2.5 nm which showed low toxicity, excellent luminescence, good photostability, and up-conversion properties.[23]
Modification
Being a new type of fluorescent nanoparticles, applications of CQD lie in the field of bioimaging and biosensing due to their biological and environmental friendly composition and excellent biocompatibility.[1] In order to survive the competition with conventional semiconductor quantum dots, a high quantum yield should be achieved. Although a good example of CQDs with ~80% quantum yield was synthesized,[29] most of the quantum dots synthesized have a quantum yield below 10% so far.[7] Surface-passivation and doping methods for modifications are usually applied for improving quantum yield.
To prevent surfaces of CQDs from being polluted by their environment, surface passivation is performed to alleviate the detrimental influence of surface contamination on their optical properties.[30] A thin insulating layer is formed to achieve surface passivation via the attachment of polymeric materials on CQDs surface treated by acid.[7]
In addition to surface passivation, doping is also a common method used to tune the properties of CQDs. Various doping methods with elements such as N,[31] S,[32] P[33] have been demonstrated for tuning the properties of CQDs, among which N doping is the most common way due to its great ability in improving the photo luminescence emissions.[34] The mechanisms by which Nitrogen doping enhances the fluorescence quantum yield of CQDs, as well as the structure of heavily N-doped CDs, are very debated issues in the literature.[35][36] Zhou et al. applied XANES and XEOL in investigating the electronic structure and luminescence mechanism in their electrochemically produced carbon QDS and found that N doping is almost certainly responsible for the blue luminescence.[37] Synthesis of new nanocomposites based on CDs have been reported with unusual properties. For example, a new nanocomposite has been designed by using of CDs and magnetic Fe3O4 nanoparticles as precursors with nanozymetic activity.[38]
Applications
Bioimaging
CQDs can be used for bioimaging due to their fluorescence emissions and biocompatibility.[39] By injecting solvents containing CQDs into a living body, images in vivo can be obtained for detection or diagnosis purposes. One example is that organic dye-conjugated CQDs could be used as an effective fluorescent probes for H2S. The presence of H2S could tune the blue emission of the organic dye-conjugated CQDs to green. So by using a fluorescence microscope, the organic dye-conjugated CQDs were able to visualize changes in physiologically relevant levels of H2S.[7] Another example can be dual-mode bioimaging using their highly accessible surface functional groups to conjugate them via EDC-NHS chemistry.[40] Saladino et al. demonstrated the concept using MW-assisted synthesized nitrogen-doped excitation-independent CQDs. These were conjugated with rhodium nanoparticles – X-ray fluorescence contrast agents – leading to dual-mode nanohybrids with both optical and X-ray fluorescent properties. Moreover, the conjugation process not only accounts for dual-mode bioimaging but also passivates the rhodium nanoparticle surface, resulting in reduced cytotoxicity.[41]
Sensing
CQDs were also applied in biosensing as biosensor carriers for their flexibility in modification, high solubility in water, nontoxicity, good photostability, and excellent biocompatibility.[1] The biosensors based on CQD and CQs-based materials could be used for visual monitoring of cellular copper,[42] glucose,[43] pH,[44] trace levels of H2O2 [38] and nucleic acid.[45] A general example is about nucleic acid lateral flow assays. The discriminating tags on the amplicons are recognized by their respective antibodies and fluorescence signals provided by the attached CQDs.[7] More generally, the fluorescence of CQDs efficiently responds to pH,[46] local polarity,[13] and to the presence of metal ions in solution,[47] which further expands their potential for nanosensing applications,[48] for instance in the analysis of pollutants.[49]
Drug delivery
The nontoxicity and biocompatibility of CQDs enable them with broad applications in biomedicine as drug carriers, fluorescent tracers as well as controlling drug release.[50][51][52][27] This is exemplified by the use of CQDs as photosensitizers in photodynamic therapy to destroy cancer cells.[53]
Catalysis
The flexibility of functionalization with various groups CQDs makes them possible to absorb lights of different wavelengths, which offers good opportunities for applications in photocatalysis.[54] CQDs-modified P25 TiO2 composites exhibited improved photocatalytic H2 evolution under irradiation with UV-Vis. The CQDs serve as a reservoir for electrons to improve the efficiency of separating of the electron-hole pairs of P25.[55]
Optronics
CQDs possess the potential in serving as materials for dye-sensitized solar cells,[56] organic solar cells,[1] supercapacitor,[57] and light emitting devices.[58] CQDs can be used as photosensitizer in dye-sensitized solar cells and the photoelectric conversion efficiency is significantly enhanced.[59] CQD incorporated hybrid silica based sol can be used as transparent Fluorescent paint,[60]
Rocket fuels
Recently, CQDs have been employed in hybrid rocket fuels.[61]
Fingerprint recovery
CQDs are used for the enhancement of latent fingerprints.[62]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 Wang, Youfu; Hu, Aiguo (2014). "Carbon quantum dots: Synthesis, properties and applications". Journal of Materials Chemistry C 2 (34): 6921–39. doi:10.1039/C4TC00988F.
- ↑ Fernando, K. A. Shiral; Sahu, Sushant; Liu, Yamin; Lewis, William K.; Guliants, Elena A.; Jafariyan, Amirhossein; Wang, Ping; Bunker, Christopher E. et al. (2015). "Carbon Quantum Dots and Applications in Photocatalytic Energy Conversion". ACS Applied Materials & Interfaces 7 (16): 8363–76. doi:10.1021/acsami.5b00448. PMID 25845394.
- ↑ Gao, Xiaohu; Cui, Yuanyuan; Levenson, Richard M; Chung, Leland W K; Nie, Shuming (2004). "In vivo cancer targeting and imaging with semiconductor quantum dots". Nature Biotechnology 22 (8): 969–76. doi:10.1038/nbt994. PMID 15258594.
- ↑ Xu, Xiaoyou; Ray, Robert; Gu, Yunlong; Ploehn, Harry J.; Gearheart, Latha; Raker, Kyle; Scrivens, Walter A. (2004). "Electrophoretic Analysis and Purification of Fluorescent Single-Walled Carbon Nanotube Fragments". Journal of the American Chemical Society 126 (40): 12736–7. doi:10.1021/ja040082h. PMID 15469243.
- ↑ Kottam, Nagaraju; S P, Smrithi (2021-01-09). "'Luminescent carbon nanodots: Current prospects on synthesis, properties and sensing applications'". Methods and Applications in Fluorescence 9 (1): 012001. doi:10.1088/2050-6120/abc008. ISSN 2050-6120. PMID 33043896. Bibcode: 2021MApFl...9a2001K. https://doi.org/10.1088/2050-6120/abc008.
- ↑ Chan, Warren C.W; Maxwell, Dustin J; Gao, Xiaohu; Bailey, Robert E; Han, Mingyong; Nie, Shuming (2002). "Luminescent quantum dots for multiplexed biological detection and imaging". Current Opinion in Biotechnology 13 (1): 40–6. doi:10.1016/S0958-1669(02)00282-3. PMID 11849956.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Lim, Shi Ying; Shen, Wei; Gao, Zhiqiang (2015). "Carbon quantum dots and their applications". Chemical Society Reviews 44 (1): 362–81. doi:10.1039/C4CS00269E. PMID 25316556.
- ↑ Li, Yan; Zhao, Yang; Cheng, Huhu; Hu, Yue; Shi, Gaoquan; Dai, Liming; Qu, Liangti (2012). "Nitrogen-Doped Graphene Quantum Dots with Oxygen-Rich Functional Groups". Journal of the American Chemical Society 134 (1): 15–8. doi:10.1021/ja206030c. PMID 22136359.
- ↑ Ye, Ruquan; Xiang, Changsheng; Lin, Jian; Peng, Zhiwei; Huang, Kewei; Yan, Zheng; Cook, Nathan P.; Samuel, Errol L.G. et al. (2013). "Coal as an abundant source of graphene quantum dots". Nature Communications 4: 2943. doi:10.1038/ncomms3943. PMID 24309588. Bibcode: 2013NatCo...4.2943Y.
- ↑ Li, Haitao; He, Xiaodie; Kang, Zhenhui; Huang, Hui; Liu, Yang; Liu, Jinglin; Lian, Suoyuan; Tsang, ChiHimA. et al. (2010). "Water-Soluble Fluorescent Carbon Quantum Dots and Photocatalyst Design". Angewandte Chemie International Edition 49 (26): 4430–4. doi:10.1002/anie.200906154. PMID 20461744.
- ↑ Sun, Ya-Ping; Zhou, Bing; Lin, Yi; Wang, Wei; Fernando, K. A. Shiral; Pathak, Pankaj; Meziani, Mohammed Jaouad; Harruff, Barbara A. et al. (2006). "Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence". Journal of the American Chemical Society 128 (24): 7756–7. doi:10.1021/ja062677d. PMID 16771487.
- ↑ Liu, Yun; Liu, Chun-yan; Zhang, Zhi-Ying (2011). "Synthesis and surface photochemistry of graphitized carbon quantum dots". Journal of Colloid and Interface Science 356 (2): 416–21. doi:10.1016/j.jcis.2011.01.065. PMID 21306724. Bibcode: 2011JCIS..356..416L.
- ↑ 13.0 13.1 13.2 Sciortino, Alice; Marino, Emanuele; Dam, Bart van; Schall, Peter; Cannas, Marco; Messina, Fabrizio (2016). "Solvatochromism Unravels the Emission Mechanism of Carbon Nanodots". The Journal of Physical Chemistry Letters 7 (17): 3419–23. doi:10.1021/acs.jpclett.6b01590. PMID 27525451.
- ↑ Demchenko, Alexander P.; Dekaliuk, Mariia O. (2016). "The origin of emissive states of carbon nanoparticles derived from ensemble-averaged and single-molecular studies". Nanoscale 8 (29): 14057–69. doi:10.1039/C6NR02669A. PMID 27399599. Bibcode: 2016Nanos...814057D.
- ↑ Khan, Syamantak; Gupta, Abhishek; Verma, Navneet C.; Nandi, Chayan K. (2015). "Time-Resolved Emission Reveals Ensemble of Emissive States as the Origin of Multicolor Fluorescence in Carbon Dots". Nano Letters 15 (12): 8300–5. doi:10.1021/acs.nanolett.5b03915. PMID 26566016. Bibcode: 2015NanoL..15.8300K.
- ↑ Mondal, Somen; Agam, Yuval; Amdursky, Nadav (2020). "Enhanced Proton Conductivity across Protein Biopolymers Mediated by Doped Carbon Nanoparticles". Small 16 (50): 2005526. doi:10.1002/smll.202005526. PMID 33108059.
- ↑ Bhattacharya, Dipsikha; Mishra, Manish K.; De, Goutam (2017). "Carbon Dots from a Single Source Exhibiting Tunable Luminescent Colors through the Modification of Surface Functional Groups in ORMOSIL Films". Journal of Physical Chemistry C 121 (50): 28106–16. doi:10.1021/acs.jpcc.7b08039.
- ↑ Rimal, Vishal; Shishodia, Shubham; Srivastava, P.K. (2020). "Novel synthesis of high-thermal stability carbon dots and nanocomposites from oleic acid as an organic substrate". Applied Nanoscience 10 (2): 455–464. doi:10.1007/s13204-019-01178-z.
- ↑ Zhou, Jigang; Booker, Christina; Li, Ruying; Zhou, Xingtai; Sham, Tsun-Kong; Sun, Xueliang; Ding, Zhifeng (2007). "An Electrochemical Avenue to Blue Luminescent Nanocrystals from Multiwalled Carbon Nanotubes (MWCNTs)". Journal of the American Chemical Society 129 (4): 744–5. doi:10.1021/ja0669070. PMID 17243794. https://figshare.com/articles/An_Electrochemical_Avenue_to_Blue_Luminescent_Nanocrystals_from_Multiwalled_Carbon_Nanotubes_MWCNTs_/3030481.
- ↑ Zhou, Jigang (2009). "Tailoring multi-wall carbon nanotubes for smaller nanostructures". Carbon 47 (3): 829–838. doi:10.1016/j.carbon.2008.11.032.
- ↑ Zhou, Jigang (2013). "An electrochemical approach to fabricating honeycomb assemblies from multiwall carbon nanotubes". Carbon 59 (3): 130–139. doi:10.1016/j.carbon.2013.03.001.
- ↑ Peng, Hui; Travas-Sejdic, Jadranka (2009). "Simple Aqueous Solution Route to Luminescent Carbogenic Dots from Carbohydrates". Chemistry of Materials 21 (23): 5563–5. doi:10.1021/cm901593y.
- ↑ 23.0 23.1 23.2 Zhu, Hui; Wang, Xiaolei; Li, Yali; Wang, Zhongjun; Yang, Fan; Yang, Xiurong (2009). "Microwave synthesis of fluorescent carbon nanoparticles with electrochemiluminescence properties". Chemical Communications (34): 5118–20. doi:10.1039/B907612C. PMID 20448965. https://semanticscholar.org/paper/e097c9df1497f763f3a566f633d7d233056bf996.
- ↑ Phadke, Chinmay; Mewada, Ashmi; Dharmatti, Roopa; Thakur, Mukeshchand; Pandey, Sunil; Sharon, Madhuri (2015). "Biogenic Synthesis of Fluorescent Carbon Dots at Ambient Temperature Using Azadirachta indica (Neem) gum". Journal of Fluorescence 25 (4): 1103–7. doi:10.1007/s10895-015-1598-x. PMID 26123675.
- ↑ Oza, Goldie; Oza, Kusum; Pandey, Sunil; Shinde, Sachin; Mewada, Ashmi; Thakur, Mukeshchand; Sharon, Maheshwar; Sharon, Madhuri (2014). "A Green Route Towards Highly Photoluminescent and Cytocompatible Carbon dot Synthesis and its Separation Using Sucrose Density Gradient Centrifugation". Journal of Fluorescence 25 (1): 9–14. doi:10.1007/s10895-014-1477-x. PMID 25367312.
- ↑ Mewada, Ashmi; Pandey, Sunil; Shinde, Sachin; Mishra, Neeraj; Oza, Goldie; Thakur, Mukeshchand; Sharon, Maheshwar; Sharon, Madhuri (2013). "Green synthesis of biocompatible carbon dots using aqueous extract of Trapa bispinosa peel". Materials Science and Engineering: C 33 (5): 2914–7. doi:10.1016/j.msec.2013.03.018. PMID 23623114.
- ↑ 27.0 27.1 Thakur, Mukeshchand; Pandey, Sunil; Mewada, Ashmi; Patil, Vaibhav; Khade, Monika; Goshi, Ekta; Sharon, Madhuri (2014). "Antibiotic Conjugated Fluorescent Carbon Dots as a Theranostic Agent for Controlled Drug Release, Bioimaging, and Enhanced Antimicrobial Activity". Journal of Drug Delivery 2014: 282193. doi:10.1155/2014/282193. PMID 24744921.
- ↑ Thakur, Mukeshchand; Mewada, Ashmi; Pandey, Sunil; Bhori, Mustansir; Singh, Kanchanlata; Sharon, Maheshwar; Sharon, Madhuri (2016). "Milk-derived multi-fluorescent graphene quantum dot-based cancer theranostic system". Materials Science and Engineering: C 67: 468–77. doi:10.1016/j.msec.2016.05.007. PMID 27287144.
- ↑ Zhu, Shoujun; Meng, Qingnan; Wang, Lei; Zhang, Junhu; Song, Yubin; Jin, Han; Zhang, Kai; Sun, Hongchen et al. (2013). "Highly Photoluminescent Carbon Dots for Multicolor Patterning, Sensors, and Bioimaging". Angewandte Chemie International Edition 52 (14): 3953–7. doi:10.1002/anie.201300519. PMID 23450679.
- ↑ Nicollian, E. H. (1971). "Surface Passivation of Semiconductors". Journal of Vacuum Science and Technology 8 (5): S39–S49. doi:10.1116/1.1316388. Bibcode: 1971JVST....8S..39N.
- ↑ Xu, Yang; Wu, Ming; Liu, Yang; Feng, Xi-Zeng; Yin, Xue-Bo; He, Xi-Wen; Zhang, Yu-Kui (2013). "Nitrogen-Doped Carbon Dots: A Facile and General Preparation Method, Photoluminescence Investigation, and Imaging Applications". Chemistry - A European Journal 19 (7): 2276–83. doi:10.1002/chem.201203641. PMID 23322649.
- ↑ Sun, Dong; Ban, Rui; Zhang, Peng-Hui; Wu, Ge-Hui; Zhang, Jian-Rong; Zhu, Jun-Jie (2013). "Hair fiber as a precursor for synthesizing of sulfur- and nitrogen-co-doped carbon dots with tunable luminescence properties". Carbon 64: 424–34. doi:10.1016/j.carbon.2013.07.095.
- ↑ Prasad, K. Sudhakara; Pallela, Ramjee; Kim, Dong-Min; Shim, Yoon-Bo (2013). "Microwave-Assisted One-Pot Synthesis of Metal-Free Nitrogen and Phosphorus Dual-Doped Nanocarbon for Electrocatalysis and Cell Imaging". Particle & Particle Systems Characterization 30 (6): 557–64. doi:10.1002/ppsc.201300020.
- ↑ Ayala, Paola; Arenal, Raul; Loiseau, Annick; Rubio, Angel; Pichler, Thomas (2010). "The physical and chemical properties of heteronanotubes". Reviews of Modern Physics 82 (2): 1843. doi:10.1103/RevModPhys.82.1843. Bibcode: 2010RvMP...82.1843A.
- ↑ Messina, F.; Sciortino, L.; Popescu, R.; Venezia, A. M.; Sciortino, A.; Buscarino, G.; Agnello, S.; Schneider, R. et al. (2016). "Fluorescent nitrogen-rich carbon nanodots with an unexpected β-C3N4nanocrystalline structure". Journal of Materials Chemistry C 4 (13): 2598–605. doi:10.1039/C5TC04096E.
- ↑ Zhou, Juan; Yang, Yong; Zhang, Chun-Yang (2013). "A low-temperature solid-phase method to synthesize highly fluorescent carbon nitride dots with tunable emission". Chemical Communications 49 (77): 8605–7. doi:10.1039/C3CC42266F. PMID 23749222.
- ↑ Zhou, Jigang; Zhou, Xingtai; Li, Ruying; Sun, Xueliang; Ding, Zhifeng; Cutler, Jeffrey; Sham, Tsun-Kong (2009). "Electronic structure and luminescence center of blue luminescent carbon nanocrystals". Chemical Physics Letters 474 (4–6): 320–324. doi:10.1016/j.cplett.2009.04.075. Bibcode: 2009CPL...474..320Z.
- ↑ 38.0 38.1 Yousefinejad, Saeed; Rasti, Hamid; Hajebi, Mehdi; Kowsari, Masoud; Sadravi, Shima; Honarasa, Fatemeh (2017). "Design of C-dots/Fe3O4 magnetic nanocomposite as an efficient new nanozyme and its application for determination of H2O2 in nanomolar level". Sensors and Actuators B: Chemical 247 (August): 691–6. doi:10.1016/j.snb.2017.02.145.
- ↑ Oza, Goldie; Ravichandran, M.; Merupo, Victor-Ishrayelu; Shinde, Sachin; Mewada, Ashmi; Ramirez, Jose Tapia; Velumani, S.; Sharon, Madhuri et al. (2016). "Camphor-mediated synthesis of carbon nanoparticles, graphitic shell encapsulated carbon nanocubes and carbon dots for bioimaging". Scientific Reports 6: 21286. doi:10.1038/srep21286. PMID 26905737. Bibcode: 2016NatSR...621286O.
- ↑ Kilic, Nüzhet Inci (2021). Graphene Quantum Dots as Fluorescent and Passivation Agents for Multimodal Bioimaging. http://urn.kb.se/resolve?urn=urn:nbn:se:kth:diva-298302.
- ↑ Saladino, Giovanni M.; Kilic, Nuzhet I.; Brodin, Bertha; Hamawandi, Bejan; Yazgan, Idris; Hertz, Hans M.; Toprak, Muhammet S. (September 2021). "Carbon Quantum Dots Conjugated Rhodium Nanoparticles as Hybrid Multimodal Contrast Agents" (in en). Nanomaterials 11 (9): 2165. doi:10.3390/nano11092165. ISSN 2079-4991. PMID 34578481.
- ↑ Zhu, Anwei; Qu, Qiang; Shao, Xiangling; Kong, Biao; Tian, Yang (2012). "Carbon-Dot-Based Dual-Emission Nanohybrid Produces a Ratiometric Fluorescent Sensor for InVivo Imaging of Cellular Copper Ions". Angewandte Chemie International Edition 51 (29): 7185–9. doi:10.1002/anie.201109089. PMID 22407813.
- ↑ Shi, Wenbing; Wang, Qinlong; Long, Yijuan; Cheng, Zhiliang; Chen, Shihong; Zheng, Huzhi; Huang, Yuming (2011). "Carbon nanodots as peroxidase mimetics and their applications to glucose detection". Chemical Communications 47 (23): 6695–7. doi:10.1039/C1CC11943E. PMID 21562663.
- ↑ Shi, Wen; Li, Xiaohua; Ma, Huimin (2012). "A Tunable Ratiometric pH Sensor Based on Carbon Nanodots for the Quantitative Measurement of the Intracellular pH of Whole Cells". Angewandte Chemie International Edition 51 (26): 6432–5. doi:10.1002/anie.201202533. PMID 22644672.
- ↑ Li, Hailong; Zhang, Yingwei; Wang, Lei; Tian, Jingqi; Sun, Xuping (2011). "Nucleic acid detection using carbon nanoparticles as a fluorescent sensing platform". Chemical Communications 47 (3): 961–3. doi:10.1039/C0CC04326E. PMID 21079843.
- ↑ Kong, Weiguang; Wu, Huizhen; Ye, Zhenyu; Li, Ruifeng; Xu, Tianning; Zhang, Bingpo (2014). "Optical properties of pH-sensitive carbon-dots with different modifications". Journal of Luminescence 148: 238–42. doi:10.1016/j.jlumin.2013.12.007. Bibcode: 2014JLum..148..238K.
- ↑ Chaudhary, Savita; Kumar, Sandeep; Kaur, Bhawandeep; Mehta, S. K. (2016). "Potential prospects for carbon dots as a fluorescence sensing probe for metal ions". RSC Advances 6 (93): 90526–36. doi:10.1039/C6RA15691F. Bibcode: 2016RSCAd...690526C.
- ↑ Bogireddy, Naveen Kumar Reddy; Barba, Victor; Agarwal, Vivechana (2019). "Nitrogen-Doped Graphene Oxide Dots-Based "Turn-OFF" H2O2, Au(III), and "Turn-OFF–ON" Hg(II) Sensors as Logic Gates and Molecular Keypad Locks". ACS Omega 4 (6): 10702–10713. doi:10.1021/acsomega.9b00858. PMID 31460168.
- ↑ Cayuela, Angelina; Laura Soriano, M.; Valcárcel, Miguel (2013). "Strong luminescence of Carbon Dots induced by acetone passivation: Efficient sensor for a rapid analysis of two different pollutants". Analytica Chimica Acta 804: 246–51. doi:10.1016/j.aca.2013.10.031. PMID 24267089.
- ↑ Mewada, Ashmi; Pandey, Sunil; Thakur, Mukeshchand; Jadhav, Dhanashree; Sharon, Madhuri (2014). "Swarming carbon dots for folic acid mediated delivery of doxorubicin and biological imaging". Journal of Materials Chemistry B 2 (6): 698–705. doi:10.1039/C3TB21436B. PMID 32261288.
- ↑ Pandey, Sunil; Mewada, Ashmi; Thakur, Mukeshchand; Tank, Arun; Sharon, Madhuri (2013). "Cysteamine hydrochloride protected carbon dots as a vehicle for the efficient release of the anti-schizophrenic drug haloperidol". RSC Advances 3 (48): 26290–6. doi:10.1039/C3RA42139B. Bibcode: 2013RSCAd...326290P.
- ↑ Pandey, Sunil; Thakur, Mukeshchand; Mewada, Ashmi; Anjarlekar, Dhanashree; Mishra, Neeraj; Sharon, Madhuri (2013). "Carbon dots functionalized gold nanorod mediated delivery of doxorubicin: Tri-functional nano-worms for drug delivery, photothermal therapy and bioimaging". Journal of Materials Chemistry B 1 (38): 4972–82. doi:10.1039/C3TB20761G. PMID 32261087.
- ↑ Juzenas, Petras; Kleinauskas, Andrius; George Luo, Pengju; Sun, Ya-Ping (2013). "Photoactivatable carbon nanodots for cancer therapy". Applied Physics Letters 103 (6): 063701. doi:10.1063/1.4817787. Bibcode: 2013ApPhL.103f3701J.
- ↑ Kim, Jinhyun; Lee, Sahng Ha; Tieves, Florian; Choi, Da Som; Hollmann, Frank; Paul, Caroline E.; Park, Chan Beum (15 October 2018). "Biocatalytic C=C Bond Reduction through Carbon Nanodot‐Sensitized Regeneration of NADH Analogues". Angewandte Chemie International Edition 57 (42): 13825–13828. doi:10.1002/anie.201804409. PMID 30062834.
- ↑ Mandal, Tapas K.; Parvin, Nargish (2011). "Rapid Detection of Bacteria by Carbon Quantum Dots". Journal of Biomedical Nanotechnology 7 (6): 846–8. doi:10.1166/jbn.2011.1344. PMID 22416585.
- ↑ Xie, Shilei; Su, Hua; Wei, Wenjie; Li, Mingyang; Tong, Yexiang; Mao, Zongwan (2014). "Remarkable photoelectrochemical performance of carbon dots sensitized TiO2 under visible light irradiation". Journal of Materials Chemistry A 2 (39): 16365–8. doi:10.1039/C4TA03203A.
- ↑ Zhu, Yirong; Ji, Xiaobo; Pan, Chenchi; Sun, Qingqing; Song, Weixin; Fang, Laibing; Chen, Qiyuan; Banks, Craig E. (2013). "A carbon quantum dot decorated RuO2 network: Outstanding supercapacitances under ultrafast charge and discharge". Energy & Environmental Science 6 (12): 3665–75. doi:10.1039/C3EE41776J.
- ↑ Zhang, Xiaoyu; Zhang, Yu; Wang, Yu; Kalytchuk, Sergii; Kershaw, Stephen V.; Wang, Yinghui; Wang, Peng; Zhang, Tieqiang et al. (2013). "Color-Switchable Electroluminescence of Carbon Dot Light-Emitting Diodes". ACS Nano 7 (12): 11234–41. doi:10.1021/nn405017q. PMID 24246067.
- ↑ Ma, Zheng; Zhang, Yong-Lai; Wang, Lei; Ming, Hai; Li, Haitao; Zhang, Xing; Wang, Fang; Liu, Yang et al. (2013). "Bioinspired Photoelectric Conversion System Based on Carbon-Quantum-Dot-Doped Dye–Semiconductor Complex". ACS Applied Materials & Interfaces 5 (11): 5080–4. doi:10.1021/am400930h. PMID 23668995.
- ↑ Mishra, Manish Kr; Chakravarty, Amrita; Bhowmik, Koushik; De, Goutam (2015). "Carbon nanodot–ORMOSIL fluorescent paint and films". Journal of Materials Chemistry C 3 (4): 714–9. doi:10.1039/C4TC02140A.
- ↑ Oztan, Cagri; Ginzburg, Eric; Akin, Mert; Zhou, Yiqun; Leblanc, Roger M.; Coverstone, Victoria (2021). "3D printed ABS/paraffin hybrid rocket fuels with carbon dots for superior combustion performance". Combustion and Flame 225: 428–434. doi:10.1016/j.combustflame.2020.11.024. https://www.researchgate.net/publication/346433516.
- ↑ Fernandes, Diogo; Krysmann, Marta J.; Kelarakis, Antonios (2015). "Carbon dot based nanopowders and their application for fingerprint recovery". Chemical Communications 51 (23): 4902–4905. doi:10.1039/C5CC00468C. PMID 25704392.
Further reading
- Bourlinos, Athanasios B.; Stassinopoulos, Andreas; Anglos, Demetrios; Zboril, Radek; Karakassides, Michael; Giannelis, Emmanuel P. (2008). "Surface Functionalized Carbogenic Quantum Dots". Small 4 (4): 455–8. doi:10.1002/smll.200700578. PMID 18350555. Bibcode: 2008APS..MARY30007B.
- Li, Haitao; He, Xiaodie; Liu, Yang; Huang, Hui; Lian, Suoyuan; Lee, Shuit-Tong; Kang, Zhenhui (2011). "One-step ultrasonic synthesis of water-soluble carbon nanoparticles with excellent photoluminescent properties". Carbon 49 (2): 605–9. doi:10.1016/j.carbon.2010.10.004.
- Zong, Jie; Zhu, Yihua; Yang, Xiaoling; Shen, Jianhua; Li, Chunzhong (2011). "Synthesis of photoluminescent carbogenic dots using mesoporous silica spheres as nanoreactors". Chem. Commun. 47 (2): 764–6. doi:10.1039/C0CC03092A. PMID 21069221.
- Krysmann, Marta J.; Kelarakis, Antonios; Dallas, Panagiotis; Giannelis, Emmanuel P. (2012). "Formation Mechanism of Carbogenic Nanoparticles with Dual Photoluminescence Emission". Journal of the American Chemical Society 134 (2): 747–50. doi:10.1021/ja204661r. PMID 22201260.
- Chandra, Sourov; Patra, Prasun; Pathan, Shaheen H.; Roy, Shuvrodeb; Mitra, Shouvik; Layek, Animesh; Bhar, Radhaballabh; Pramanik, Panchanan et al. (2013). "Luminescent S-doped carbon dots: An emergent architecture for multimodal applications". Journal of Materials Chemistry B 1 (18): 2375–82. doi:10.1039/C3TB00583F. PMID 32261072.
- Kim, Jinhyun; Lee, Sahng Ha; Tieves, Florian; Choi, Da Som; Hollmann, Frank; Paul, Caroline E.; Park, Chan Beum (15 October 2018). "Biocatalytic C=C Bond Reduction through Carbon Nanodot‐Sensitized Regeneration of NADH Analogues". Angewandte Chemie International Edition 57 (42): 13825–13828. doi:10.1002/anie.201804409. PMID 30062834.


