Chemistry:Meso-Stilbene dibromide
From HandWiki
Short description: Organic molecule
| |
| Names | |
|---|---|
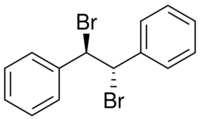
| Preferred IUPAC name
1,1′-[(1R,2S)-1,2-Dibromoethane-1,2-diyl]dibenzene | |
| Other names
Stilbene dibromide; meso-1,2-dibromo-1,2-diphenyl ethane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C14H12Br2 | |
| Molar mass | 340.058 g·mol−1 |
| Appearance | White solid |
| Melting point | 241 °C (466 °F; 514 K)[2] |
| 0.4-0.9[1] | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H302, H314 | |
| P260, P264, P270, P280, P301+312, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P321, P330, P332+313, P337+313, P362, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
meso-Stilbene dibromide is an organic compound with a formula of (C6H5CH(Br))2.[3] It is one of three isomeric stilbene dibromides, the others being the pair of enantiomers. All are white solids.[4]
Synthesis and reactions
meso-Stilbene dibromide can be prepared by treatment of (E)-stilbene with bromine.[5]
Reaction of stilbene dibromide with base gives diphenylacetylene.[4]
References
- ↑ Weissberger, A. (1945). "Dipole Moment and Structure of Organic Compounds. XVII.[1] The Electric Moments of α- and β-Stilbene Dibromide and of p-Diacetylbenzene". J. Am. Chem. Soc. 67 (5): 778–779. doi:10.1021/ja01221a025.
- ↑ Kulangiappar, K.; Ramaprakash, M.; Vasudevan, D.; Raju, T. (2016). "Electrochemical bromination of cyclic and acyclic enes using biphase electrolysis". Synthetic Communications 46 (2): 145–153. doi:10.1080/00397911.2015.1125498. https://figshare.com/articles/journal_contribution/1617199.
- ↑ Schroth, Werner; Zeitschrift für Chemie 1977, V17(2), P56 CAPLUS
- ↑ 4.0 4.1 Smith, Lee Irvin; Falkov, M. M. (1942). "Diphenylacetylene". Org. Synth. 22: 50. doi:10.15227/orgsyn.022.0050.
- ↑ Gilbert, J.C.; Martin, S.F. (2002). Experimental Organic Chemistry: A Miniscale and Macroscale Approach, third edition.. London: Thomson. pp. 348–353. ISBN 978-0-03-034048-2.
 |

