Chemistry:4,6-Dimethyldibenzothiophene
From HandWiki
| |
| Names | |
|---|---|
| Other names
4,6-DMDBT
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C14H12S | |
| Molar mass | 212.31 g·mol−1 |
| Appearance | white solid |
| Density | 1.18 g/cm3 |
| Melting point | 153–157 °C (307–315 °F; 426–430 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302 | |
| P264, P270, P301+317Script error: No such module "Preview warning".Category:GHS errors, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
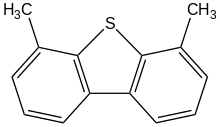
4,6-Dimethyldibenzothiophene is an organosulfur compound with the formula (C
6H
3CH
3)
2S. It is one of several dimethyl derivatives of benzothiophene. The compound is of particular interest as an organosulfur contaminant in petroleum that is recalcitrant. Both methyl groups shield the sulfur center from desulfurization.[2][3]
The compound can be oxidized to the sulfoxide with hydrogen peroxide.[4] Many methods for synthesis of this compound have been described. Often the methods proceed via derivatives of diphenyl sulfide.[5]
References
- ↑ "4,6-Dimethyldibenzothiophene" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/1268103#section=Safety-and-Hazards.
- ↑ Song, Chunshan; Ma, Xiaoliang (2003). "New Design Approaches to Ultra-Clean Diesel Fuels by Deep Desulfurization and Deep Dearomatization". Applied Catalysis B: Environmental 41 (1–2): 207–238. doi:10.1016/s0926-3373(02)00212-6.
- ↑ Gates, B.C.; Topsøe, Henrik (1997). "Reactivities in Deep Catalytic Hydrodesulfurization: Challenges, Opportunities, and the Importance of 4-Methyldibenzothiophene and 4,6-Dimethyldibenzothiophene". Polyhedron 16 (18): 3213–3217. doi:10.1016/s0277-5387(97)00074-0.
- ↑ Sato, Kazuhiko; Hyodo, Mamoru; Aoki, Masao; Zheng, Xiao-Qi; Noyori, Ryoji (2001). "Oxidation of Sulfides to Sulfoxides and Sulfones with 30% Hydrogen Peroxide under Organic Solvent- and Halogen-Free Conditions". Tetrahedron 57 (13): 2469–2476. doi:10.1016/s0040-4020(01)00068-0.
- ↑ Meille, Valérie; Schulz, Emmanuelle; Lemaire, Marc; Faure, René; Vrinat, Michel (1996). "An Efficient Synthesis of Pure 4,6-Dimethyldibenzothiophene". Tetrahedron 52 (11): 3953–3960. doi:10.1016/S0040-4020(96)00041-5.
 |

