Biology:RyhB
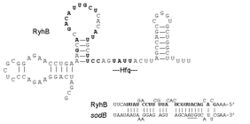
RyhB RNA is a 90 nucleotide RNA that down-regulates a set of iron-storage and iron-using proteins when iron is limiting; it is itself negatively regulated by the ferric uptake repressor protein, Fur (Ferric uptake regulator).
Discovery
The gene was independently identified in two screens, named RyhB by Wassarman et al. and called SrI by Argaman et al. and was found to be expressed only in stationary phase.[2][3]
Function and regulation
RyhB RNA levels are inversely correlated with mRNA levels for the sdhCDAB operon, encoding succinate dehydrogenase, as well as five other genes previously shown to be positively regulated by Fur by an unknown mechanism. These include two other genes encoding enzymes in the tricarboxylic acid cycle, acnA and fumA, two ferritin genes, ftnA and bfr, and a gene for superoxide dismutase, sodB.[4] A number of other genes have been predicted computationally and verified as targets by microarray analysis: napF, sodA, cysE, yciS, acpS, nagZ and dadA.[1] RyhB is bound by the Hfq protein, that increases its interaction with its target messages.
A comparative genomics target prediction approach suggests that the mRNAs of eleven additional iron containing proteins are controlled by RyhB in Escherichia coli. Two of those (erpA, nirB) and two additional targets that are not directly related to iron (nagZ, marA) were verified with a GFP reporter system.[5][6]
It has been shown that RyhB has a role in targeting the polycistronic transcript iscRSUA for differential degradation. RyhB binds to the second cistron of iscRSUA, which encodes machinery for biosynthesis of Fe-S clusters. This binding promotes cleavage of the downstream iscSUA transcript. This cleavage leaves the 5′ IscR transcript which is a transcriptional regulator responsible regulating several genes that depend on cellular Fe-S level.[7]
More recent data indicate a potential dual function role for RyhB. In this capacity it may act both as an RNA-RNA interaction based regulator and as a transcript encoding for a small protein.[8]
RyhB is analogous to PrrF RNA found in Pseudomonas aeruginosa,[9] to HrrF RNA in Haemophilus species [10] and to IsaR1 in cyanobacteria.[11][12]
First sRNA shown to mediate persistence to antibiotics in E.coli. The finding may lead to discovery of novel treatments for persistent infections.[13]
Naming
The RyhB gene name is an acronym composed of R for RNA, y for unknown function (after the protein naming convention), with the h representing the ten-minute-interval section of the E. coli map the gene is found in. The B comes from the fact that this was one of two RNA genes identified in this interval.[3] Other RNAs using this nomenclature include RydB RNA, RyeB RNA, RyeE RNA and RyfA RNA.
References
- ↑ 1.0 1.1 "Target prediction for small, noncoding RNAs in bacteria". Nucleic Acids Research 34 (9): 2791–2802. 2006. doi:10.1093/nar/gkl356. PMID 16717284.
- ↑ "Novel small RNA-encoding genes in the intergenic regions of Escherichia coli". Current Biology 11 (12): 941–950. June 2001. doi:10.1016/S0960-9822(01)00270-6. PMID 11448770.
- ↑ 3.0 3.1 "Identification of novel small RNAs using comparative genomics and microarrays". Genes & Development 15 (13): 1637–1651. July 2001. doi:10.1101/gad.901001. PMID 11445539.
- ↑ "A small RNA regulates the expression of genes involved in iron metabolism in Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America 99 (7): 4620–4625. April 2002. doi:10.1073/pnas.032066599. PMID 11917098. Bibcode: 2002PNAS...99.4620M.
- ↑ "Comparative genomics boosts target prediction for bacterial small RNAs". Proceedings of the National Academy of Sciences of the United States of America 110 (37): E3487–3496. September 2013. doi:10.1073/pnas.1303248110. PMID 23980183. Bibcode: 2013PNAS..110E3487W.
- ↑ "Translational control and target recognition by Escherichia coli small RNAs in vivo". Nucleic Acids Research 35 (3): 1018–1037. 2007. doi:10.1093/nar/gkl1040. PMID 17264113.
- ↑ "Small RNA-induced differential degradation of the polycistronic mRNA iscRSUA". The EMBO Journal 28 (11): 1551–1561. June 2009. doi:10.1038/emboj.2009.116. PMID 19407815.
- ↑ "Differentiation of ncRNAs from small mRNAs in Escherichia coli O157:H7 EDL933 (EHEC) by combined RNAseq and RIBOseq - ryhB encodes the regulatory RNA RyhB and a peptide, RyhP". BMC Genomics 18 (1): 216. February 2017. doi:10.1186/s12864-017-3586-9. PMID 28245801.
- ↑ "Identification of tandem duplicate regulatory small RNAs in Pseudomonas aeruginosa involved in iron homeostasis". Proceedings of the National Academy of Sciences of the United States of America 101 (26): 9792–9797. June 2004. doi:10.1073/pnas.0403423101. PMID 15210934. Bibcode: 2004PNAS..101.9792W.
- ↑ "HrrF is the Fur-regulated small RNA in nontypeable Haemophilus influenzae". PLOS ONE 9 (8): e105644. 2014-01-01. doi:10.1371/journal.pone.0105644. PMID 25157846. Bibcode: 2014PLoSO...9j5644S.
- ↑ "Acclimation of Oxygenic Photosynthesis to Iron Starvation Is Controlled by the sRNA IsaR1". Current Biology 27 (10): 1425–1436.e7. May 2017. doi:10.1016/j.cub.2017.04.010. PMID 28479323. https://pearl.plymouth.ac.uk/bitstream/handle/10026.1/9292/UoP_Deposit_Agreement%20v1.1%2020160217.pdf?sequence=2&isAllowed=y.
- ↑ "The iron-stress activated RNA 1 (IsaR1) coordinates osmotic acclimation and iron starvation responses in the cyanobacterium Synechocystis sp. PCC 6803". Environmental Microbiology 20 (8): 2757–2768. February 2018. doi:10.1111/1462-2920.14079. PMID 29468839.
- ↑ "Escherichia coli by Reducing Cellular Metabolism". Frontiers in Microbiology 9: 136. 2018. doi:10.3389/fmicb.2018.00136. PMID 29467745.
Further reading
- "A small RNA promotes siderophore production through transcriptional and metabolic remodeling". Proceedings of the National Academy of Sciences of the United States of America 107 (34): 15223–15228. August 2010. doi:10.1073/pnas.1007805107. PMID 20696910. Bibcode: 2010PNAS..10715223S.
- "Induction of the ferritin gene (ftnA) of Escherichia coli by Fe(2+)-Fur is mediated by reversal of H-NS silencing and is RyhB independent". Molecular Microbiology 75 (3): 637–657. February 2010. doi:10.1111/j.1365-2958.2009.06977.x. PMID 20015147.
- "Impact of the small RNA RyhB on growth, physiology and heterologous protein expression in Escherichia coli". FEMS Microbiology Letters 275 (2): 221–228. October 2007. doi:10.1111/j.1574-6968.2007.00880.x. PMID 17784860.
- "The small RNA RyhB activates the translation of shiA mRNA encoding a permease of shikimate, a compound involved in siderophore synthesis". Molecular Microbiology 64 (5): 1260–1273. June 2007. doi:10.1111/j.1365-2958.2007.05733.x. PMID 17542919.
- "RyhB, an iron-responsive small RNA molecule, regulates Shigella dysenteriae virulence". Infection and Immunity 75 (7): 3470–3477. July 2007. doi:10.1128/IAI.00112-07. PMID 17438026.
- "Control of Fur synthesis by the non-coding RNA RyhB and iron-responsive decoding". The EMBO Journal 26 (4): 965–975. February 2007. doi:10.1038/sj.emboj.7601553. PMID 17268550.
- "RyhB small RNA modulates the free intracellular iron pool and is essential for normal growth during iron limitation in Escherichia coli". Molecular Microbiology 62 (4): 1181–1190. November 2006. doi:10.1111/j.1365-2958.2006.05439.x. PMID 17078818.
- "Fur regulates acid resistance in Shigella flexneri via RyhB and ydeP". Molecular Microbiology 58 (5): 1354–1367. December 2005. doi:10.1111/j.1365-2958.2005.04920.x. PMID 16313621.
- "Effect of RyhB small RNA on global iron use in Escherichia coli". Journal of Bacteriology 187 (20): 6962–6971. October 2005. doi:10.1128/JB.187.20.6962-6971.2005. PMID 16199566.
- "Characterization of Vibrio cholerae RyhB: the RyhB regulon and role of ryhB in biofilm formation". Infection and Immunity 73 (9): 5706–5719. September 2005. doi:10.1128/IAI.73.9.5706-5719.2005. PMID 16113288.
- "Characterization of the small untranslated RNA RyhB and its regulon in Vibrio cholerae". Journal of Bacteriology 187 (12): 4005–4014. June 2005. doi:10.1128/JB.187.12.4005-4014.2005. PMID 15937163.
- "Micros for microbes: non-coding regulatory RNAs in bacteria". Trends in Genetics 21 (7): 399–404. July 2005. doi:10.1016/j.tig.2005.05.008. PMID 15913835.
- "Both RNase E and RNase III control the stability of sodB mRNA upon translational inhibition by the small regulatory RNA RyhB". Nucleic Acids Research 33 (5): 1678–1689. 2005. doi:10.1093/nar/gki313. PMID 15781494.
- "Iron-responsive bacterial small RNAs: variations on a theme". Metallomics 5 (4): 276–286. April 2013. doi:10.1039/c3mt20224k. PMID 23340911.
- "The Vibrio parahaemolyticus small RNA RyhB promotes production of the siderophore vibrioferrin by stabilizing the polycistronic mRNA". Journal of Bacteriology 195 (16): 3692–3703. August 2013. doi:10.1128/JB.00162-13. PMID 23772063.
- Cascales, Eric, ed (2013). "The sRNA RyhB regulates the synthesis of the Escherichia coli methionine sulfoxide reductase MsrB but not MsrA". PLOS ONE 8 (5): e63647. doi:10.1371/journal.pone.0063647. PMID 23671689. Bibcode: 2013PLoSO...863647B.
- "Rapid degradation of Hfq-free RyhB in Yersinia pestis by PNPase independent of putative ribonucleolytic complexes". BioMed Research International 2014: 798918. 2014. doi:10.1155/2014/798918. PMID 24818153.
- "Constitutive expression of RyhB regulates the heme biosynthesis pathway and increases the 5-aminolevulinic acid accumulation in Escherichia coli". FEMS Microbiology Letters 350 (2): 209–215. January 2014. doi:10.1111/1574-6968.12322. PMID 24188714.
- "The small RNA RyhB contributes to siderophore production and virulence of uropathogenic Escherichia coli". Infection and Immunity 82 (12): 5056–5068. December 2014. doi:10.1128/IAI.02287-14. PMID 25245805.
- "The small RNA RyhB homologs from Salmonella typhimurium participate in the response to S-nitrosoglutathione-induced stress". Biochemical and Biophysical Research Communications 450 (1): 641–645. July 2014. doi:10.1016/j.bbrc.2014.06.031. PMID 24937451.
External links
 |
