Chemistry:2-Pinanol
From HandWiki
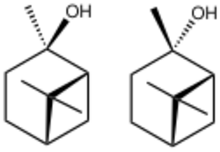
| |
| Names | |
|---|---|
| Other names
2,6,6-trimethylbicyclo[3.1.1]heptan-2-ol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H18O | |
| Molar mass | 154.253 g·mol−1 |
| Appearance | colorless solid |
| Melting point | 78–79 °C (cis) 58–59 °C (trans |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H302, H311, H312, H315, H319 | |
| P264, P264+265Script error: No such module "Preview warning".Category:GHS errors, P270, P280, P301+317Script error: No such module "Preview warning".Category:GHS errors, P302+352, P305+351+338, P316Script error: No such module "Preview warning".Category:GHS errors, P317Script error: No such module "Preview warning".Category:GHS errors, P321, P330, P332+317Script error: No such module "Preview warning".Category:GHS errors, P337+317Script error: No such module "Preview warning".Category:GHS errors, P361+364Script error: No such module "Preview warning".Category:GHS errors, P362+364Script error: No such module "Preview warning".Category:GHS errors, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
2-Pinanol is a collection of bicyclic terpenoid derived from the terpene pinene, but containing a tertiary alcohol. Both cis and trans isomers exist. Both are chiral They are produced by deoxygenation of corresponding cis- and trans-2-pinane hydroperoxide, which in turn can be produced by autoxidation of pinane with air.[2] Heating 2-pinanol gives linalool.[3]
References
- ↑ "2-Pinanol" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/10128#section=Safety-and-Hazards.
- ↑ Erman, Mark B.; Kane, Bernard J. (2008). "Chemistry Around Pinene and Pinane: A Facile Synthesis of Cyclobutanes and Oxatricyclo-Derivative of Pinane from cis- and trans-Pinanols". Chemistry & Biodiversity 5 (6): 910–919. doi:10.1002/cbdv.200890104. PMID 18618388.
- ↑ Eggersdorfer, Manfred (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a26_205.
 |
