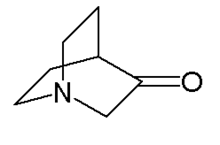
Chemistry:3-Quinuclidone
From HandWiki
| |
| Names | |
|---|---|
| Other names
1-azabicyclo[2.2.2]octan-3-one
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C7H11NO | |
| Molar mass | 125.171 g·mol−1 |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H302, H312, H332, H411 | |
| P261, P264, P270, P271, P273, P280, P301+317Script error: No such module "Preview warning".Category:GHS errors, P302+352, P304+340, P317Script error: No such module "Preview warning".Category:GHS errors, P321, P330, P362+364Script error: No such module "Preview warning".Category:GHS errors, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
3-Quinuclidinone is a bicyclic organic compounds with chemical formula HC(C
2H
4)
2(C(O)CH
2)N. Its basicity is indicated by the pKa of the conjugate acid, which is 7.2. In contrast quinuclidine is about 100x more basic.[2]
Synthesis and reactions
Its hydrochloride salt can be synthesized by a Dieckman condensation:[3] It is a precursor to quinuclidine.

Organic reduction of 3-quinuclidone gives the compound quinuclidine, structurally related to DABCO, which has one additional bridgehead nitrogen atom.
References
- ↑ "Quinuclidin-3-one" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/19507#section=Safety-and-Hazards.
- ↑ Aggarwal, Varinder K.; Emme, Ingo; Fulford, Sarah Y. (2003). "Correlation between pKa and Reactivity of Quinuclidine-Based Catalysts in the Baylis−Hillman Reaction: Discovery of Quinuclidine as Optimum Catalyst Leading to Substantial Enhancement of Scope". The Journal of Organic Chemistry 68 (3): 692–700. doi:10.1021/jo026671s. PMID 12558387.
- ↑ H. U. Daeniker, C. A. Grob (1964). "3-Quinuclidone Hydrochloride". Organic Syntheses 44: 86. doi:10.15227/orgsyn.044.0086.
 |
