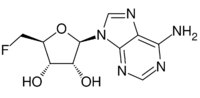
Chemistry:5'-Deoxy-5'-fluoroadenosine
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
5′-Deoxy-5′-fluoroadenosine
| |
| Systematic IUPAC name
(2R,3R,4S,5S)-2-(6-Amino-9H-purin-9-yl)-5-(fluoromethyl)oxolane-3,4-diol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H12FN5O3 | |
| Molar mass | 269.236 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
5′-Deoxy-5′-fluoroadenosine is the first step in the biosynthesis of organic fluorides. It is synthesized by the fluorinase catalyzed addition of a fluoride ion to S-adenosyl-L-methionine, releasing L-methionine as a by product.[1] Purine nucleoside phosphorylase mediates a phosphorolytic cleavage of the adenine base to generate 5-fluoro-5-deoxy-D-ribose-1-phosphate.
References
- ↑ "Biochemistry: biosynthesis of an organofluorine molecule". Nature 416 (6878): 279. 2002. doi:10.1038/416279a. PMID 11907567. Bibcode: 2002Natur.416..279O.
 |

