Chemistry:ACT-539313
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| Drug class | Orexin receptor antagonist |
| Pharmacokinetic data | |
| Elimination half-life | 3.3–6.5 hours[1][2] |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| UNII | |
| Chemical and physical data | |
| Formula | C23H23N7O2 |
| Molar mass | 429.484 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
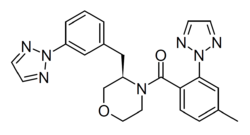
ACT-539313 is an orexin antagonist medication which is under development for the treatment of binge eating disorder and was previously under development for the treatment of anxiety disorders.[4][5][6][7][1][2] It is an orally active small-molecule compound with an elimination half-life of 3.3 to 6.5 hours and acts as a selective orexin OX1 receptor antagonist (1-SORA).[4][1][2] As of May 2022, the drug is in phase 2 clinical trials for binge eating disorder.[4] Following negative efficacy results of a phase 2 trial of ACT-539313 for binge eating disorder, Idorsia (the developer of ACT-539313) signaled in May 2022 that it would not pursue further development of the drug for this indication.[8]
References
- ↑ 1.0 1.1 1.2 1.3 "First-in-human study with ACT-539313, a novel selective orexin-1 receptor antagonist". British Journal of Clinical Pharmacology 86 (7): 1377–1386. July 2020. doi:10.1111/bcp.14251. PMID 32067262. "ACT-539313 ((4-methyl-2-[1,2,3]triazol-2-yl-phenyl)-[(R)- 3-(3-[1,2,3]triazol-2-yl-benzyl)-morpholin-4-yl]-methanone) is an orally active, reversible, selective OX1 receptor antagonist (1-SORA) that readily crosses the blood–brain barrier.".
- ↑ 2.0 2.1 2.2 "Multiple-dose clinical pharmacology of the selective orexin-1 receptor antagonist ACT-539313". Progress in Neuro-Psychopharmacology & Biological Psychiatry 108: 110166. June 2021. doi:10.1016/j.pnpbp.2020.110166. PMID 33159976.
- ↑ "Orexin Signaling: A Complex, Multifaceted Process". Frontiers in Cellular Neuroscience 16: 812359. 2022. doi:10.3389/fncel.2022.812359. PMID 35496914.
- ↑ 4.0 4.1 4.2 "ACT 539313 - AdisInsight". https://adisinsight.springer.com/drugs/800049623.
- ↑ "Hypocretins (orexins): The ultimate translational neuropeptides". Journal of Internal Medicine 291 (5): 533–556. May 2022. doi:10.1111/joim.13406. PMID 35043499.
- ↑ "Counterbalanced microcircuits for Orx1 and Orx2 regulation of stress reactivity". Medicine in Drug Discovery 8: 100059. December 2020. doi:10.1016/j.medidd.2020.100059. ISSN 2590-0986.
- ↑ "Experimental Drugs for Panic Disorder: An Updated Systematic Review". Journal of Experimental Pharmacology 13: 441–459. 2021. doi:10.2147/JEP.S261403. PMID 33889031.
- ↑ "Just months after its first FDA approval, Idorsia dumps binge-eating drug candidate after PhII fail". https://endpts.com/just-months-after-its-first-fda-approval-idorsia-dumps-binge-eating-drug-candidate-after-phii-fail/.
