Chemistry:Amonafide
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Trade names | Xanafide, Quinamed |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
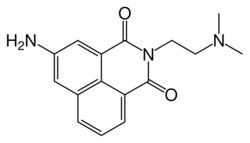
| Formula | C16H17N3O3 |
| Molar mass | 299.330 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Amonafide (originally AS1413) (INN, trade names Quinamed and Xanafide) was a drug that was being studied in the treatment of cancer. It belongs to a novel family of chemotherapeutic drugs called Naphthalimides and is a potential topoisomerase inhibitor and DNA intercalator.[1][2]
It was being developed as an anti-cancer therapy by Antisoma.[3]
As of 2008[update], it is in Phase III clinical trials. e.g. In March 2010 it is Phase III trial against secondary acute myeloid leukaemia (AML).[4] In June 2010, it gained an FDA Fast Track Status for the treatment of Secondary Acute Myeloid Leukaemia.[citation needed]
See also
References
- ↑ "Amonafide: a potential role in treating acute myeloid leukemia". Expert Opinion on Investigational Drugs 20 (7): 995–1003. July 2011. doi:10.1517/13543784.2011.585756. PMID 21591994.
- ↑ "Amonafide: a future in treatment of resistant and secondary acute myeloid leukemia?". Expert Review of Hematology 5 (1): 17–26. February 2012. doi:10.1586/ehm.11.68. PMID 22272701.
- ↑ "Antisoma's Shares Plummet 70% as Cancer Candidate Bombs in Phase III NSCLC Trial". Genetic Engineering & Biotechnology News. 29 March 2010. http://www.genengnews.com/news/bnitem.aspx?name=78979622.
- ↑ Clinical trial number NCT00715637 for "Phase III Randomized Study of Amonafide (AS1413) and Cytarabine Versus Daunorubicin and Cytarabine in Patients With Secondary Acute Myeloid Leukemia (AML)- the ACCEDE Study" at ClinicalTrials.gov
External links
- Amonafide entry in the public domain NCI Dictionary of Cancer Terms
- Clinical trials of amonafide at ClinicalTrials.gov
![]() This article incorporates public domain material from the U.S. National Cancer Institute document "Dictionary of Cancer Terms".
This article incorporates public domain material from the U.S. National Cancer Institute document "Dictionary of Cancer Terms".
 |
